nabilop234
10.01.2021 •
Chemistry
What's the theoretical yield if 5.50 g.of Hydrogen react with nitrogen to from amonia? 3H2+N2=2NH3
Solved
Show answers
More tips
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
Answers on questions: Chemistry
- S Social Studies I need general information about Mesoamerican civilization pleasee a couple of bullets would really help :)...
- M Mathematics Factorization of X ^ 4 + Y to the power 4 minus 3 x to the power 2 y to thepower 2...
- B Biology How do living things use each type of nucleic acid...
- S Social Studies Which political system has a central supreme government and holds supreme authority over its local government units?...

Ответ:
The theoretical yield : 31.11 g
Further explanationGiven
Reaction
3H2+N2=2NH3
Required
the theoretical yield
Solution
mol of Hydrogen :mol = mass : Ar
mol = 5.5 g : 2
mol = 2.75
mol of Ammonia :From the equation, mol ratio of H₂ : NH₃ = 3 : 2, so mol NH₃ =
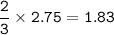
Mass Ammonia (MW=17 g/mol):mass =mol x MW
mass = 1.83 x 17
mass= 31.11 g
Ответ:
From top to bottom electropositivity increases
Left to right it reduces
Explanation:
Electropositivity is also known as metallicity. It is a measure of the tendency of atoms of an element to lose electrons. It is closely related to the ionization energy and indeed the electronegativity.
The lower the ionization energy, the more electropositive a substance is. Across the period from left to right, electropositivity or metallicity decreases and from top to down a group, it increases.