davis52202
02.10.2019 •
Chemistry
What stressors will affect the value for keq according to le chatelier's principle?
a. increasing the concentration
b. decreasing the concentration
c. decreasing the pressure
d. increasing the temperature
Solved
Show answers
More tips
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
Answers on questions: Chemistry
- M Mathematics 52% of u.s adults have very little confidence in newspapers. You randomly select 10 u.s adults who have very little confidence in news papers is (a) exactly five, (b) at least six,...
- E English What is the most likely purpose of describing the amphitheater as an agent of poetic justice? to suggest that the king is a fair and just ruler to reveal that the trials always...
- S SAT If you are convicted of dui, your fine and jail time will be increased if ....
- E English I need to write a short Gothic horror story for my English essay (500+ words) I just need a plot for my story. Thanks...
- B Biology What weather factor is also a measurement of energy held within the air? a.humidity b.pressure c.temperature d.wind...

Ответ:
Option (D) is the correct answer.
Explanation:
According to Le Chatelier's principle, any disturbance caused in an equilibrium reaction will tend to shift the equilibrium in a direction away from the disturbance.
So, any change in concentration, pressure, catalyst etc will not lead to any change in equilibrium constant.
But when we increase temperature then equilibrium will shift in a direction where temperature is reduced again.
For example,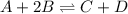
So, when we increase the temperature in forward direction then equilibrium will shift in the backward direction leading to more of A and B and less of C and D.
Thus, we can conclude that increasing temperature stressors will affect the value for according to Le Chatelier's principle.
according to Le Chatelier's principle.
Ответ:
Write chemical formula of Methanol,
CH₃-OH
Find out Subscripts:
There is only one subscript (₃) written after Hydrogen, it means there are 3 Hydrogen atoms attached to Carbon atoms.
Add all atoms in molecule:
Carbons = 1
Hydrogens = 4
Oxygens = 1
Total Atoms
6 Atoms