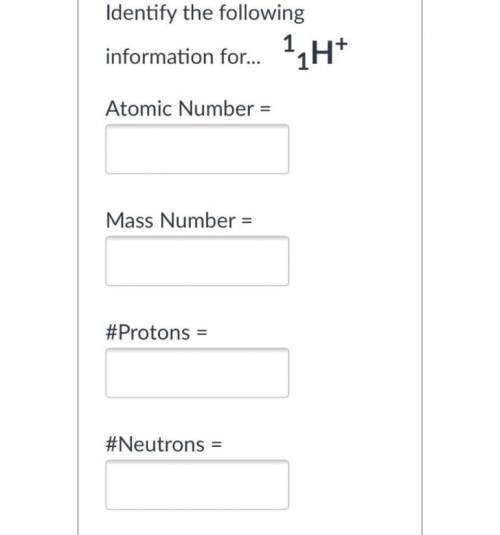
What the atomic number
what is the mass number
#protons ? and neutrons and electrons?
Solved
Show answers
More tips
- A Auto and Moto Which alarm system to choose?...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
Answers on questions: Chemistry
- C Chemistry Cho 32g CuO tác dụng vừa đủ với H2SO4 a, Tìm khối lượng của H2SO4 b, Tìm khối lượng của CuSO4 tạo ra sau phản ứng...
- C Chemistry 1) An insoluble, nonreactive metal sphere weighing 16.924g is added to 21.7mL of water in a graduated cylinder. The water level rises to 38.9. Calculate the density of...
- C Chemistry What levels are normal in the Toxins homeostasis...
- C Chemistry Naturally occurring gallium consists of two isotopes. One of those isotopes is 71Ga with an isotopic mass of 70.9247050 amu and an abundance of 39.892% . What is the mass...
- C Chemistry What is photolytic decomposition reaction ???...
- C Chemistry What determines an atom s atomic number?...
- C Chemistry Which statements are true of a basic solution? check all that apply. a. to make it more concentrated, add base b. to make it more dilute, add base c. to make it more dilute,...
- C Chemistry Glucose, c6h12o6,c6h12o6, is used as an energy source by the human body. the overall reaction in the body is described by the equation c6h12o6(aq) 6o2(g)⟶6co2(g) 6h2o(l)...
- C Chemistry When the following reaction is completed and written in the form of a net ionic equation, which of the following elements will NOT be present in the equation? Pb(NO3)2(aq)...
- C Chemistry How do you know what to...

Ответ:
Explanation:
Atomic number = 1
Mass number = 1
Proton = 1
Neutron = mass number - atomic number
= 1 - 1
= 0
Ответ:
Representation of chemical reactivity by symbols follows two absolute rules:
Explanation:
1
.
Mass is conserved.
2
.
Charge is conserved.
What do we mean by this? If we start with 10 g of reactant FROM ALL SOURCES, AT MOST we can get 10 g of product; and in practice we are not even going to get that. Since mass is a fundamental property of atoms and molecules it follows that atoms and molecules are conserved in EVERY chemical reaction.
)
Because it is not balanced (why not?), we can reject it out of hand, because we know that it does not reflect reality.
)
80 g of reactant gives 80 g of product
...charge and mass are balanced here. And so this is a reasonable representation of reality.
I know I am being absolute when I say that charge and mass are conserved, but this reflects EVERY CHEMICAL REACTION, EVERY EXPERIMENT EVER PERFORMED: when a reaction is studied in detail,
garbage out has always equalled garbage in.
And how do we know that masses are conserved; that stoichiometry operates? How else but by experiment? How else but by extensive and quantitative study of particular chemical reactions?
Every chemical reaction ever performed (and as far we know, TO BE PERFORMED), displays conservation of mass. And today we have a particle view of chemical reactivity, and our ideas, developed over only some 200-300 years, insist that matter is conserved. That molecules and atoms themselves have discrete masses, which are certainly measurable, supports our notion of conservation of mass.
This idea can be extended to the representation of redox reactions, where we can invoke the electron as a charged particle that is exchanged between species in a redox process. Charge is conserved, as well as mass.
If this all seems a lot to take in, remember you will only be asked to balance equations to which you have already been introduced. Alkanes combust in air to give carbon dioxide and water, iron combines with oxygen to give rust,
If you can write a chemical equation that balances mass and charge, you have proposed a reasonable chemical pathway. See here for redox equations.
Explanation: