What usually happens to the rate of reaction when the concentration of one of the reactants is increased
Solved
Show answers
More tips
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry The mass of a proton is 1.67×10 ^ −27kg. what is the mass of a proton in picograms?...
- C Chemistry Consider the balanced reaction bellow. how many moles of barium hydroxide,ba(oh)2, would be required to react with 117g hydrogen bromide,2hbr+ba(oh)2=babr2+2h2...
- C Chemistry Write the electronic configuration and draw the atomic orbital diagram from 1 to 7...
- C Chemistry Which structural formula correctly represents an organic compound...
- C Chemistry What do periods on the periodic table represent?...
- C Chemistry 24 what is the definition of a covalent bond? a. a bond between two positive ions b. a bond between two atoms c. a bond between two negative ions d. a bond between a positive...
- B Business Preparation of the statement of cash flows does not involve: Select one: a. Computing the net increase or decrease in cash. b. Computing and reporting net cash provided or used...
- B Business The plaintiff is a State A corporation that entered into a contract with the defendant under which the defendant agreed to manufacture and sell equipment to the plaintiff. The...
- M Mathematics The tourism department of a certain country would like to decide which projects to fund during the coming year. The projects were divided into three main categories: religious,...
- M Mathematics sand falls from an overhead bin and accumulates in a conical pile with a radius that is always four times its height. suppose the height of the pile infcreases at a rate of 1cm/s...

Ответ:
The rate of the reaction increases because more collisions can occur.
Ответ:
Further Explanation:
Stoichiometry:
Theamountof species present in the reaction is determined with the help of stoichiometryby the relationship between reactants and products. It is used to determine the moles of a chemical species when moles of other chemical species present in the reaction is given.
Balanced chemical reaction between sodium carbonate and sulphuric acid is as follows:
According to stoichiometry of reaction, one mole of reacts with one mole of
reacts with one mole of 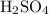 to produce one mole of
to produce one mole of 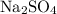 , one mole of
, one mole of 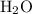 and one mole of
and one mole of 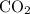 . So stoichiometric ratio between
. So stoichiometric ratio between  and
and 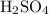 is 1:1.
is 1:1.
The formula to calculate number of moles of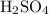 is as follows:
is as follows:
Substitute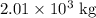 for mass of
for mass of 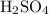 and 98.079 g/mol for molar mass of
and 98.079 g/mol for molar mass of 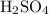 in equation (1).
in equation (1).
Since one mole of reacts with one mole of
reacts with one mole of 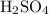 , 20493.7 moles of
, 20493.7 moles of 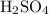 reacts with 20493.7 moles of
reacts with 20493.7 moles of  .
.
The formula to calculate mass of is as follows:
is as follows:
Substitute 20493.7 mol for moles of and 105.9888 g/mol for molar mass of
and 105.9888 g/mol for molar mass of  in equation (2).
in equation (2).
Learn more:
Calculate the moles of chlorine in 8 moles of carbon tetrachloride: link Calculate the moles of ions in the solution: linkAnswer details:
Grade: Senior School
Subject: Chemistry
Chapter: Mole concept
Keywords: stoichiometry, Na2CO3, H2SO4, Na2SO4, CO2, H2O, balanced chemical reaction, 1:1, one mole, stoichiometric ratio, 2172.1 kg.