aniyahrhs1213
28.05.2021 •
Chemistry
What volume of water would you add to 35.81 mL of 1.59 M acetic acid solution to make it a 0.97 M?
Solved
Show answers
More tips
- A Auto and Moto What to expect from the new VW Golf 7?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
Answers on questions: Chemistry
- C Chemistry 2H2O(l) + energy à 2H2(g) + O2(g) Which of the following phrases best describes this reaction?...
- C Chemistry To prepare 250mL of calcium chloride solution with a molar concentration of 1.20mol/L, what mass of calcium chloride would be required?...
- C Chemistry A “happy” atom is one that has a valence orbital. octet rule...
- C Chemistry Potassium chloride (KCI) and sucrose (C12H22011) both dissolve in water. Which solution is able to conduct electricity?...
- C Chemistry What is the process by which a solute forms a solution in a solvent....
- C Chemistry Using the Debye-Huckel equation, the activity coefficient for H+ in the aqueous solution of 0.0250 M sodium chloride is:...
- C Chemistry A 4.05 mol sample of Kr has a volume of 311 mL. How many moles of Kr are in a 6.55 L sample at the same temperature and pressure...
- C Chemistry Which organisms are a part of Protista? select all correct answers. []protozoa []amoeba []algae []virus Help ASAP!...
- C Chemistry Student 10You are supplied with a stock aqueous hydrobromic acid solution of concentration 0.834 M, and you pipet 25.00 mL of the acid into a 250 mL Erlenmeyer flask. The...
- C Chemistry Question 7.Water decomposes into hydrogen and oxvgen during electrolysis. A sample of water is electrolysed.The hydrogen and oxygen are collected separately. After ten minutes,...

Ответ:
A volume of 58.69 mL of water would you add to 35.81 mL of 1.59 M acetic acid solution to make it a 0.97 M.
Explanation:
Given: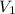 = 35.81 mL,
= 35.81 mL,  = 1.59 M
= 1.59 M
Formula used to calculate volume is as follows.
Substitute the values into above formula as follows.
Thus, we can conclude that a volume of 58.69 mL of water would you add to 35.81 mL of 1.59 M acetic acid solution to make it a 0.97 M.
Ответ:
the last one, It must have both male and female parts in the flower.
Explanation: