19thomasar
26.06.2019 •
Chemistry
What would be the temperature change 3.0 g of water absorbed 15 j of heat
Solved
Show answers
More tips
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry A 679-mL sample of unknown HCl solution reacts completely with Na2CO3 to form 11.1 g CO2. What was the concentration of the HCl solution?...
- C Chemistry Question 3 (1 point) The Theory of Natural Selection teaches us that the organisms with the best traits tend to thrive and produce more offspring with each new generation. True O...
- C Chemistry How many moles of Mg are needed to produce 8.3 moles of MgO?...
- C Chemistry Help pls i would really appreciate it...
- C Chemistry the lack of. safeguards that were clearly created and established in the criminal justice for adults was not followed in the juvenile system due to the wide application of the concept...
- C Chemistry Why are hydrogen bonds the strongest of the intermolecular forces....
- C Chemistry Which atmospheric gas has the greatest effect on the weather conditions associated with an air mass? 1. oxygen 2. nitrogen 3. methane 4. water vapor...
- C Chemistry Chlorine is located in Group 17, Period 3. Which is chlorine most likely to be? O A. A soft, shiny, highly reactive metal B. A metalloid with properties of both metals and nonmetals...
- C Chemistry Que sentido ha tenido y tiene hoy la filosofía?...
- C Chemistry Please answer quickly...

Ответ:
1.2 °C
Step-by-step explanation:
The formula relating the heat (q) absorbed by an object, the mass (m), and the temperature change (ΔT) is
q = mCΔT Divide both sides by mC
ΔT = q/(mC)
Data:
q = 15 J
m = 3.0 g
C = 4.184 J·°C⁻¹g⁻¹
Calculation:
ΔT = 15/(3.0 × 4.184)
ΔT = 15/12.6
ΔT = 1.2 °C
Ответ:
The change in temperature of the sample has been .
.
The specific heat has been defined as the amount of heat required to raise the temperature of 1 gram of substance by 1 degree Celsius.
The expression for specific heat has been given as:

Computation for Change in TemperatureFor the given water sample,
The mass of sample has been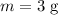
The heat absorbed by the sample has been
The specific heat of water has been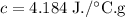
Substituting the values for change in temperature, :
:
The change in temperature of the sample has been .
.
Learn more about specific heat here:
link
Ответ:
hi
Explanation: