trashbinkid
18.07.2019 •
Chemistry
When 2.50 mol of mg3n2 are allowed to react how many moles of h2o also react?
Solved
Show answers
More tips
- H Health and Medicine How to Lower Your Child s Temperature?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- P Philosophy 8 привычек, чтобы достичь счастливой жизни...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- H Health and Medicine How to Choose the Right Glasses?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
Answers on questions: Chemistry
- C Chemistry Erosion never plz help...
- C Chemistry How many elements un 2c3h5o2...
- C Chemistry MULTIPLE CHOICE True or False: Any metal object that contains iron, such as steel, will be attracted to a magnet....
- C Chemistry The solution to a system of linear equations is (-3, -3). Which system of linear equations has this point as its solution? I m being timed!...
- C Chemistry Help ! i don’t know how to calculate this , it’s due at 11:59 pm...
- C Chemistry As a professor Daniel must complete a research project each year in order to stay on staff. Daniel is an engineer and has to deal with many complicated mathematical calculations....
- C Chemistry What is 3 x 10^4 multiplied by 5 x 10^2?...
- C Chemistry What can be best shown in these investigations even though different methods were used?...
- C Chemistry Una muestra de 1.5276 g de CdCl2 se convirtió a cadmio metálico y productos libres de cadmio mediante un proceso electrolítico. El peso del cadmio metálico fue de 0.9367 g. Si se...
- C Chemistry What is the value of the reaction quotient, Q, for the voltaic cell constructed from the following two half-reactions when the Zn2 concentration is 0.0100 M and the Ag concentration...

Ответ:
Notice that in the equation given, there is 1 mol of Mg3N2 and 6 mol of H2O. Consequently, the question is requesting, if we took 2.5 mol of Mg3N2, how many mol of H2O would be essential to complete a similar reaction -- in quintessence, multiplying everything by 2.5:
the solution would be: 6 * 2.5 = 15 mol
Ответ:
28.144 g of![Al_2O_3[tex] will be produced.Explanation: We are given that Al reacts with [tex]Fe_2O_3](/tpl/images/0056/6875/2d242.png) , then the balanced chemical reaction is:
, then the balanced chemical reaction is:
It is given that is present in excess, so it is an excess reagent and Al is the limiting reagent.
is present in excess, so it is an excess reagent and Al is the limiting reagent.
Now, 2 moles of Al produces 1 mole of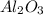 , which means that (2×27g) = 54g of Al produces 102g of
, which means that (2×27g) = 54g of Al produces 102g of 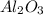 .
.
Therefore, 14.9g of Al will produce =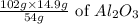
= 28.144g of