When 4.98 g of NaOH was dissolved in 52.79 g of water in a calorimeter at 23.7 oC, the temperature of the solution went up to 50.1 oC. What is the enthalpy change in kJ/mole of sodium hydroxide? Assume the specific heat of the mixture is the same as water.
Solved
Show answers
More tips
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry What is the variable that a scientist changes when conducting an experiment...
- C Chemistry What new scientific evidence led to changes in dalton s atomic theory?...
- C Chemistry Match element to how many valence electrons it has al s na si mg p 4 5 3 6 2...
- C Chemistry A compound, Y of sodium forms a white powder. It is a constituent of baking powder and isused in some antacids. When heated it gives a compound, Y which is anhydrous and absorbs...
- C Chemistry Help i will mark brainliest...
- C Chemistry Calculate the relative rate of diffusion of : H2 (molar mass 2.0 g/mol) compared to that of 2H2 (molar mass 4.0 g/mol) and the relative rate of diffusion of O2 (molar mass 32...
- C Chemistry What is the term for an object s strength of motion? O Momentum O Acceleration O Force O Velocity...
- H History I will mark you brainlest What is the first step in the naturalization process? taking an oath getting fingerprinted taking a civics test filling out an application...
- M Mathematics WHAT S THE WORD 10-Point Puzzle Find a common English 3-letter word, knowing that: LEG has no common letter with it. ERG has one common letter, not at the correct place. SIR...
- M Mathematics Please answer correctly...

Ответ:
Change in enthalpy is -51.072 kJ/mol.
Explanation:
Molar mass of NaOH = 39.997 g/mol
So, 4.98 g of NaOH =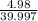 moles of NaOH = 0.125 moles of NaOH
moles of NaOH = 0.125 moles of NaOH
Total mass of solution = (4.98+52.79) g = 57.77 g
Heat consumed by solution = , where m is mass C is specific heat and
, where m is mass C is specific heat and  is change in temperature.
is change in temperature.
So, heat consumed by solution =![[57.77g\times 4.186\frac{J}{g.^{0}\textrm{C}}\times (50.1-23.7)^{0}\textrm{C}]](/tpl/images/0616/0525/cdabc.png)
= 6384 J
It is an exothermic process as temperature increases during dissolution. Hence change in enthalpy should be negative.
Change in enthalpy = -(heat consumed by solution)/(no. of moles of NaOH)
=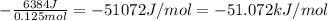
Ответ: