hernandez09297
11.07.2019 •
Chemistry
When a 3.00 grams sample of a compound containing only c, h, and o was completely burned, 1.17 grams of h2o and 2.87 grams of co2 were formed. what is the simplest formula of the compound?
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- F Family and Home What is Most Important in Men s Lives?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry 1,400L of nitrogen gas at a pressure of 1.25 atm has its pressure changed to 17.5 atm. What will be its new volume at the new pressure? (Note, no temp given for either...
- C Chemistry 9. Compound A, C5H11Br, when heated with alcoholic NaOH, gave compound B, C5H10. Compound B decolorized bromine (in CCl4) to give C. B also gave compound D when treated...
- C Chemistry A steel pin has a mass of 5.2 grams and a volume of 0.65 cubic centimeters. Calculate its density...
- C Chemistry 1. an automobile changes energy into different forms. the total amount of energy does not change due to the...
- H History Hostility towards immigrants by the native population is called: A.nativism. B.monopoly. C.migration. D.corporation....
- E English 3. If you were to draw a web outline about the most important people in a story, what word would most likely go in the middle? web conflict character plot...
- M Mathematics 3.5.PS-10 Aaliyah compares the heights of two plants to see which plant grows more per day. The table shows the height of Plant 1. in centimeters, over 5 days. The graph...
- E English Can someone give me some ideas of hooks that could be used to write about this picture?...
- M Mathematics Daddy please help a + b = b + a UwU...
- H History What did the Sedition Act do?...

Ответ:
-
Solution:- From given masses of carbon dioxide and water we could calculate the moles that helps to calculate the moles of C and H.
Molar mass of carbon dioxide = 44 gram per mol
molar mass of water = 18.02 gram per mol
From given info, combustion of compound gives 1.17 grams of water and 2.87 grams of carbon dioxide. Let's calculate the moles of these:
=
Similarly,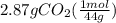
=
One mol of water has two moles of H. So, the moles of H would be two times the moles of water as calculated above.
So, moles of H = 2* 0.0649 = 0.1298 mol
One mol of carbon dioxide contains one mol of C. So, the moles of C would be equal to the moles of carbon dioxide calculated above.
moles of C = 0.0652 mol
Let's convert the moles of H and C to grams so that we could calculate the amount of oxygen present in the sample as:
grams of H in sample = 1.008 x 0.1298 = 0.1308 g
grams of C in sample = 12*0.0652 = 0.7824 g
If we subtract the sum of the masses of C and H from sample mass then it would give as the mass of oxygen since the sample has only C, H and O.
mass of O in sample = 3.00g - (0.1308 g + 0.7824 g)
= 3.00 g - 0.9132 g
= 2.0868 g
Let's convert these grams of oxygen to moles on dividing by it's atomic mass as:
= 0.130 mol O
Now, we have the moles of all the three atoms and we know that an empirical formula is the simplest whole number ratio of the moles of atoms. So, let's calculate the ratio. For this, we divide the moles of each by the least one of them.Looking at the moles, the least value is for carbon. So, let's divide the moles of each by the moles of C as:
C =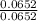 = 1
= 1
H = = 2
= 2
O = = 2
= 2
The ratio of C, H and O is 1:2:2. So, the simplest formula of the compound is .
.
Ответ:
[The rate differential results from induced fit on the enzyme upon binding of glucose. This conformational change excludes water from gaining access to the -phosphate group on ATP, and brings the same group close to the OH group on carbon 6 of glucose]
Explanation: