caitlynnpatton1208
09.03.2020 •
Chemistry
When ammonium nitrate is added to a suspension of magnesium hydroxide in water, the Mg(OH)2 dissolves. Write a net ionic equation to show how this occurs. Do not include physical states and use the smallest possible integer coefficients.
Solved
Show answers
More tips
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- G Goods and services What Useful Foods Can You Buy at Supermarkets?...
- F Food and Cooking How to Determine Healthy, Nutritious Food for Yourself?...
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- W Work and Career Can Skill Alone Make You a Professional?...
- F Family and Home Daughter says: If you don t want to do it, don t do it. Should we persuade her?...
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
Answers on questions: Chemistry
- C Chemistry How do water molecules behave?...
- C Chemistry Which pH corresponds to the equivalence point for the graph below? 14 pH 12 10 8 6 4 2 0 100 0 20 40 60 80 Volume of Base Added (mL)...
- C Chemistry A solution of an acid could have a pH of __* 3 to 5 6 to 8 9 to 11 12 to 14...
- C Chemistry Which multimedia does this ad from the Centers for Disease Control’s advertising campaign for the flu vaccine include? (more than one)A.) animationB.) imageC.) textD.) graphE.)...
- C Chemistry The water level in a graduated cylinder stands at 20.0 ml before and at 26.2 ml after a 16.74 g metal sample is lowered into the cylinder. what is the density of the sample....
- C Chemistry How manhhy neutrons does element x have if its atomic number is 24 and its mass number is 77? ?...
- C Chemistry The aluminum foil on a certain roll has a total area of 18.5 m squared and a mass of 1275 g. using density of 2.7 g per cubic centimeter for aluminum, determine the thickness...
- C Chemistry How many electrons are in the highest energy level of sulfur? a. 2 b. 8 c. 6 d. 4...
- C Chemistry How will you ensure the amount of sun energy is transferred each container...
- C Chemistry 5 How many particles are present in a 84 gm of sample of Barium hydroxide? If you can please show the work...

Ответ:
Net ionic: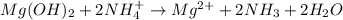
Explanation:
Due to this acid-base reaction,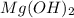 become soluble whenever
become soluble whenever 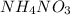 is added to suspension of
is added to suspension of 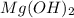 .
.
In this reaction,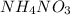 acts as an acid and
acts as an acid and 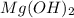 acts as a base.
acts as a base.
Molecular equation: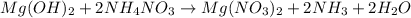
Total ionic: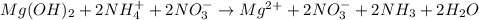
Net ionic: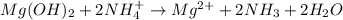
Ответ:
Total Long Term Liabilities = 3449000 USD
Explanation:
Let's sort out the data given in order to make it more presentable.
Accounts:
5-year Bonds Payable 9% = 3000000 USD
Interest Payable = 52000 USD
Premium on Bonds Payable = 99000 USD
Notes Payable (3 months.) = 42000 USD
Notes Payable (5 yr.) = 163000 USD
Mortgage Payable ($13000 due currently) = 200000 USD
Salaries and wages Payable = 18000 USD
Income Taxes Payable (due 3/15 of 2022) = 24000 USD
Now, we have to take the liabilities from this set of data and calculate it.
Following are the long term liabilities which will be added together to get the total sum:
1. 5-year Bonds Payable 9% = 3000000 USD
2. Premium on Bonds Payable = 99000 USD
3. Notes Payable (5 yr.) = 163000 USD
4. Mortgage Payable ($13000 due currently) = 200000 - 13000 = 187000 USD
Now, we have to add these amounts to get the total long term liabilities.
Total Long Term Liabilities = 3000000 + 99000 + 63000 + 187000
Total Long Term Liabilities = 3449000 USD