mvasquez3122p4vahv
14.06.2020 •
Chemistry
When aqueous solutions of sodium chloride and silver (I) acetate combine, what are the products?
Solved
Show answers
More tips
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Science and Technology Colliders: How They Work and Why They Matter...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...

Ответ:
The products are sodium acetate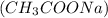 and solid silver chloride
and solid silver chloride 
Explanation:
A double displacement reaction is one in which exchange of ions take place. The salts which are soluble in water are designated by symbol (aq) and those which are insoluble in water and remain in solid form are represented by (s) after their chemical formulas.
The reaction of aqueous solutions of sodium chloride and silver (I) acetate is :
Thus the products are aqueous sodium acetate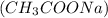 and solid silver chloride
and solid silver chloride 
Ответ: