When magnesium chloride reacts with water, 5.85 l hci(g) is produced
Solved
Show answers
More tips
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry Measuring volume in mL...
- C Chemistry Which statement defines calorimeter...
- C Chemistry Find the standard decimal form of 5x 1/10+ 7x 1/100...
- C Chemistry Identify the type of equation and write the products for the following reaction: Ba+S8...
- C Chemistry According to the following reaction, how many grams of potassium sulfate will be formed upon the complete reaction of 23.8 grams of potassium hydroxide with excess potassium hydrogen...
- C Chemistry What is true about two neutral atoms of the element gold?...
- C Chemistry What is the density of an object with a mass of 130 g and a volume of 5 cm^3?...
- B Biology What does it mean to view a plan of section...
- M Mathematics “el gráfico de la función f (x) = - 0,2x pasa por el oro gen en el plano cartesiano” verdadero o falso justifica...
- M Mathematics Pls help answer chooses a. associative property of addition b. associative property of multiplication commutative c.property of addition d. commutative property of multiplication...

Ответ:
Explanation:
Molarity is defined as the number of moles per liter of solution.
Mathematically, Molarity =
Since it is given that the molarity of a solution of 14.0 g and volume is 150 mL or 0.15 L.
Whereas number of moles =
So, molar mass of is 97.94 g/mol.
is 97.94 g/mol.
Thus, number of moles =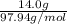
= 0.142 mol
Therefore, calculate the molarity as follows.
Molarity =
=
= 0.946 mol/L
Hence, we can conclude that molarity of the solution is 0.946 mol/L.