brookedeanovich
21.06.2021 •
Chemistry
When the following aqueous solutions are mixed together, a precipitate forms. Balance the net ionic equation in standard form for the reaction that occurs and determine the sum of the coefficients.
Sodium sulfide and silver nitrate - 3 or 4
Lead(II) nitrate and sodium chloride -3 or 4
Calcium nitrate and potassium carbonate - 3or 4
Barium nitrate and sodium hydroxide -3 or 4
Silver nitrate and sodium chloride -3 or 4
Solved
Show answers
More tips
- C Computers and Internet Why is Yandex s robot Bless.yandex.ru Important?...
- S Science and Technology How to Restore the Diamond Shine to Your Tarnished Silverware...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- L Leisure and Entertainment How to Find a Phone Number by Address: The Ultimate Guide...
- F Food and Cooking Pu-erh Tea: History, Varieties, Benefits, and Risks...
- S Style and Beauty Why is Sugaring Better than Waxing for Hair Removal?...
- C Computers and Internet How to Delete a Page on VKontakte: The Ultimate Guide from Experts...
- F Food and Cooking How to Make Delicious Plov: Secrets and Recipes...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
Answers on questions: Chemistry
- C Chemistry How many liters is 3.3 moles of a gas?...
- C Chemistry Which statement best describes the most likely temperatures of Portland, Oregon, and Eugene, Oregon, on this day in January? А Eugene was a little colder than Portland...
- C Chemistry 4 Ethanol is made by fermentation. How is ethanol obtained from the fermentation mixture? A chromatography B crystallisation c electrolysis fractional distillation...
- C Chemistry What is a disadvantage of using nuclear power to produce electricity? A. Nuclear waste must be safely stored for many years. B. Nuclear fission releases more air pollution...
- C Chemistry If I have 9.1 moles of a gas at a pressure of 6.4 atm and a volume of 8 liters, what is the temperature in Kelvin?...
- C Chemistry Help me please 18 points Your lips are more sensitive to touch than your forehead. What is different about the touch receptive fields in the lips compared to the forehead?...
- C Chemistry Julie goes to Silver River each week for 4 weeks and collects 5 samples of water to test the pH. Replication or Reptition?...
- C Chemistry 22.Oxidation occurs when materials containing are exposed to oxygen and water...
- C Chemistry Heat of fusion of ethanol 0.1 kJ/g 0.9 kJ/g Heat of vaporization of ethanol Based on the information in the table, how much heat is needed to melt 10 g of ethanol?...
- C Chemistry Question 1 If a solution contains 0.5 grams of NaCl and 100 grams of water, NaCl is the solute and water is the solvent. A) True B) False Question 2 In a solution,...

Ответ:
For (a): The balanced net ionic equation is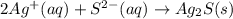 and the sum of coefficients is 4
and the sum of coefficients is 4
For (b): The balanced net ionic equation is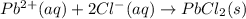 and the sum of coefficients is 4
and the sum of coefficients is 4
For (c): The balanced net ionic equation is and the sum of coefficients is
and the sum of coefficients is
For (d): The balanced net ionic equation is and the sum of coefficients is 4
and the sum of coefficients is 4
For (e): The balanced net ionic equation is and the sum of coefficients is 3
and the sum of coefficients is 3
Explanation:
Net ionic equation is defined as the equations in which spectator ions are not included.
Spectator ions are the ones that are present equally on the reactant and product sides. They do not participate in the reaction.
For (a): Sodium sulfide and silver nitrateThe balanced molecular equation is:
The complete ionic equation follows:
As sodium and nitrate ions are present on both sides of the reaction. Thus, they are considered spectator ions.
The net ionic equation follows:
Sum of the coefficients = [2 + 1 + 1] = 4
For (b): Lead(II) nitrate and sodium chlorideThe balanced molecular equation is:
The complete ionic equation follows:
As sodium and nitrate ions are present on both sides of the reaction. Thus, they are considered spectator ions.
The net ionic equation follows:
Sum of the coefficients = [2 + 1 + 1] = 4
For (c): Calcium nitrate and potassium carbonateThe balanced molecular equation is:
The complete ionic equation follows:
As potassium and nitrate ions are present on both sides of the reaction. Thus, they are considered spectator ions.
The net ionic equation follows:
Sum of the coefficients = [1 + 1 + 1] = 3
For (d): Barium nitrate and sodium hydroxideThe balanced molecular equation is:
The complete ionic equation follows:
As sodium and nitrate ions are present on both sides of the reaction. Thus, they are considered spectator ions
The net ionic equation follows:
Sum of the coefficients = [2 + 1 + 1] = 4
For (e): Silver nitrate and sodium chlorideThe balanced molecular equation is:
The complete ionic equation follows:
As sodium and nitrate ions are present on both sides of the reaction. Thus, they are considered spectator ions.
The net ionic equation follows:
Sum of the coefficients = [1 + 1 + 1] = 3
Ответ:
what
Explanation: