Which class of organic compound is commonly used in perfumes?
a. amine
b. alcohol
c. ester
d. ether
Solved
Show answers
More tips
- F Food and Cooking How to Make Delicious Plov: Secrets and Recipes...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry How can I write the equations for the following acid-base reactions and predict (by checking values) which side the equilibrium prefers? Protons to be moved will be...
- C Chemistry One element has a atomic mass of 122.211 u and the other has one of 121.537 with a relative abundance of 67.76%. Calculate the isotopic mass of the least abundant...
- C Chemistry What is the airline of Fiji...
- C Chemistry What is the airlines of Brazil...
- C Chemistry X(+1M) + (–2N) = 0 What does x have to be? (+2M) + y(–1N) = 0 What does y have to be? x(+2M) + y(–3N) = 0 What do x and y have to be? x = and y =...
- C Chemistry Which chemical agent is thought to be associated with runner s high ?...
- C Chemistry Which alcohol will undergo acid-catalyzed dehydration under the mildest conditions?...
- C Chemistry How many kilograms of sodium chloride are contained in 1.50 x 10^4 liters of a mixture of sodium chloride and water that has a density of 1.0559 g/cm^3 and is 8.00%...
- C Chemistry What can you do with this web app. What are some of the things you can do with this simulation ?...
- C Chemistry What is the daughter nucleus produced when 79Kr undergoes positron emission? daughter nucleus:...

Ответ:
Option (c) is the correct answer.
Explanation:
Esters have a chemical formula as RCOOR'.
where, R and R' can either be different or same groups. These are polar in nature.
Esters have low molecular weight and hence they are used as fragrances. Esters have a fruity smell and for this reason, they are widely used in perfumes, cosmetics, synthetic flavors etc.
Thus, we can conclude that esters are the class of organic compound is commonly used in perfumes.
Ответ:
Explanation:
From the question we are told that:
Factor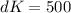
Temperature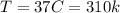
Activation energy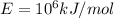
Generally the Arhenius equation is mathematically given by
Where