reneethacker20p8wdgn
10.10.2021 •
Chemistry
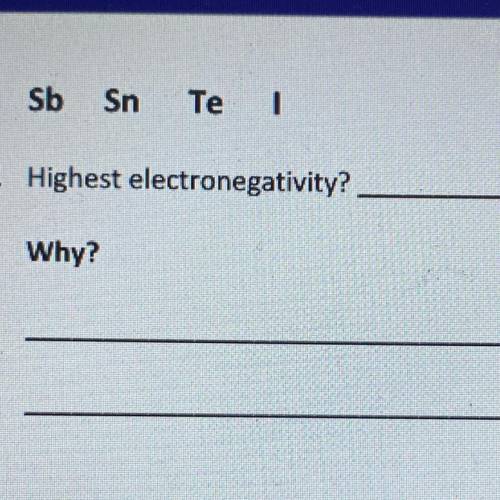
Which element has the highest electronegativity and why?
Solved
Show answers
More tips
- F Food and Cooking What Foods Can Nursing Moms Eat?...
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry Values for the molar mass of hydrogen, oxygen, and water molecules are given in the table below. What mass of water is formed when 2 moles of hydrogen react with 1 mole...
- C Chemistry What would be the volume, in liters measured at STP, of 0.56 moles of carbon monoxide?...
- C Chemistry I really need help on this...
- C Chemistry Help this is due in 10 minuets!...
- C Chemistry Cell mutations within a DNA sequence are......
- C Chemistry Calculate the number of molecules in 54,0 g of water, Show Your Work...
- C Chemistry 1. Modelaţi structura atomilor, repartizaţi electronii pe starturi, indicaţi grupa, perioada, procesul de ionizare şi caracterul chimic pentru elementele: 27 13 Al 31...
- C Chemistry Is selenium hexasulfide ionic or covalent?...
- C Chemistry Which is a renewable energy source? a. Coal b. Petroleum c. natural gas d. hydroelectric...
- C Chemistry Plants contain xylem and phloem tissues for carrying water and food. What organ system in animals performs a similar function as xylem and phloem in plants? Digestive...

Ответ:
See below
Explanation:
Hello there!
Electronegativity is the atom's tendency to attract electrons in a chemical bond
There are two trends to electronegativity:
Electronegativity increases from bottom to top in a group (Li has a greater electronegativity than Fr, for example) Electronegativity increases from left to right across a period (the further right the group, the greater the electronegativity)Looking at a period table, Sb, Sn, Te, and I are all in the same period, so we'll need to decide which element has the highest electronegativity based on the group.
Sn belongs to group 4A (group 14) Sb belongs to group 5A (group 15)Te belongs to group 6A (group 16) I belongs to group 7A (group 17)As I belongs to group 7A, the group that is the farthest right based off of the options given, I has the highest electronegativity
Hope this helps!
Ответ:
Tc
Explanation: