Which of the following has the highest concentration of h+ ions? a. 0.001 m hcl b. 0.01 m hcl c. 10 m hcl d. 0.1 m hcl
Solved
Show answers
More tips
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Which answer accurately describes the difference between speed and velocity? read each answer choice carefully. question 4 options: a. if speed is constant, then velocity...
- C Chemistry Your roommate has learned that burning approximately 3,500 food calories (the food calorie is a quantity equal to a kilocalorie, kcal) results in a weight loss of 1 lbm,...
- C Chemistry As you saw in the video, the ph of a 0.1 m solution of acetic acid, hc2h3o2, (ka ≡ 1.8 × 10−5) is 2.9. based on this observation, which statement below best describes...
- C Chemistry Which of the following would decrease the ph level of an acidic solution? (2 points) adding a base to the acid increasing the concentration of hydroxide ions decrease...
- C Chemistry What is the basis for water s high melting / freezing point?...
- C Chemistry If a neutral atom of Bromine (Br) was to gain another electron, it would have the same electron configuration as which element?...
- C Chemistry Which statement explains why some elements decay and others do not?...
- M Mathematics An insurance agent says the standard deviation of the total hospital charges for patients involved in a crash in which the vehicle struck a construction barricade is...
- H History Kumbi saleh traded primarily in gold and salt salt and copper copper and gold copper and bronze...
- M Mathematics What is the width or length of a rectangle with an area that equals 45.5cm2...

Ответ:
Answer : The correct option is, (C) 10 M HCl
Explanation :
When 1 mole of HCl dissociates then it gives 1 mole of hydrogen ion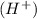 and 1 mole of chloride ion
and 1 mole of chloride ion 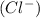 .
.
The balanced chemical reaction is,
(A) 0.001 M HCl
0.001 M concentration means, 0.001 moles present in 1 liter of solution.
When 0.001 M of HCl dissociates then it gives 0.001 of hydrogen ion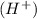 and 0.001 M of chloride ion
and 0.001 M of chloride ion 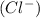 .
.
(B) 0.01 M HCl
0.01 M concentration means, 0.01 moles present in 1 liter of solution.
When 0.01 M of HCl dissociates then it gives 0.01 of hydrogen ion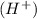 and 0.01 M of chloride ion
and 0.01 M of chloride ion 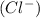 .
.
(C) 10 M HCl
10 M concentration means, 10 moles present in 1 liter of solution.
When 10 M of HCl dissociates then it gives 10 of hydrogen ion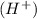 and 10 M of chloride ion
and 10 M of chloride ion 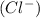 .
.
(D) 0.001 M HCl
0.1 M concentration means, 0.1 moles present in 1 liter of solution.
When 0.1 M of HCl dissociates then it gives 0.1 of hydrogen ion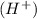 and 0.1 M of chloride ion
and 0.1 M of chloride ion 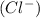 .
.
From this we conclude that, 10 M HCl has the highest concentration of hydrogen ion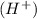 .
.
Ответ:
The equivalence point is where the number of moles of one reactant reacts completely with the moles of the other reactant.
The equivalence point is found by using suitable indicators and carrying out a titration of the acid and base. Indicators include phenolphthalein and methyl orange. Equivalence points on a pH curve is observed as a vertical line.