karsenbeau
02.12.2020 •
Chemistry
which of the following is an example of an electromagnetic wave A. an ocean wave B. A sound wave C. A light wave D. A wave traveling along a rope
Solved
Show answers
More tips
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry The graph shows tide heights from a location in Canada. Which statement is best supported by the data? Tide height varies only at high tide. The lowest high tides occurred...
- C Chemistry What is an acronym for the rainbow colors? I just want to know if anybody else knows this....
- C Chemistry What would the ionic formula be for the following ionic compound: sodium + bromine Na2Br2 NaBr2 Na2Br NaBr...
- C Chemistry In an experiment, 3.5g of element A reacted with 4.0g of element G to form a compoundCalculate the empirical formula of this compound.(relative atomic masses: A=7, G = 16)You...
- C Chemistry 1pt A geology student wants to apply the law of horizontality to determine the age of a rock taken from an area. What can the student determine about the rock based on this...
- C Chemistry Why should batteries be stored when is not being used?...
- M Mathematics Which of the following ordered pairs are solutions to the system of equations below? 3x + 2y = 9 y = 4x + 10 (-1, 6) (-1, -6) (1, 6) (1, -6)...
- M Mathematics A child is playing games with empty soda cups. There are three sizes: small, medium, and large. After some experimentation she discovered she was able to measure out 160 ounces...
- B Business Assume that the U.S. one-year interest rate is 3 percent and the one-year interest rate on Australian dollars is 6 percent. The U.S. expected annual inflation is 5 percent,...
- B Business Bond T is a zero coupon bond and has 11 years until maturity. If the yield to maturity is 10%, the Macaulay duration of this bond is...

Ответ:
Light waves
Explanation:
It isn't sound because it isn't infrared, involve electromagneticism, or light/radiation. An ocean wave is a literal wave of water, and a wave traveling along a rope is way off course. Choose a light wave. Also, brainliest would be nice. Glad to help
Ответ:
The mass percentage of nitrogen in the sample is 6.04 %
Explanation:
To calculate the number of moles for given molarity, we use the equation:
Molarity of HCl solution = 0.150 M
Volume of solution = 65.0 mL = 0.065 L (Conversion factor: 1 L = 1000 mL)
Putting values in above equation, we get:
The chemical equation for the reaction of HCl and ammonia follows:
By Stoichiometry of the reaction:
1 mole of HCl reacts with 1 mole of ammonia
So, of HCl will react with =
of HCl will react with = 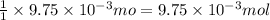 of ammonia
of ammonia
1 mole of ammonia contains 1 mole of nitrogen and 3 moles of hydrogen element
Moles of nitrogen in ammonia =
To calculate the mass for given number of moles, we use the equation:Moles of nitrogen =
Molar mass of nitrogen = 14 g/mol
Putting values in above equation, we get:

To calculate the mass percentage of nitrogen in the sample, we use the equation:Mass of sample= 2.25 g
Mass of nitrogen = 0.136 g
Putting values in above equation, we get:
Hence, the mass percentage of nitrogen in the sample is 6.04 %