daymakenna3
18.07.2019 •
Chemistry
Which of the following statements best describes a limiting reactant?
a. the reactant that is used up the least
b. the reactant that is used up the most
c. the reactant that is used up before the others
d. the reactant that is left over after the reaction
Solved
Show answers
More tips
- F Food and Cooking What Foods Can Nursing Moms Eat?...
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...

Ответ:
c. The reactant that is used up before the others
Explanation:
For example:
If we are given 100 g of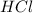 and 100 g of calcium.
and 100 g of calcium.
As can be seen from the chemical equation, 2 moles of hydrochloric acid react with 1 mole of calcium.
According to mole concept, 1 mole of every substance weighs equal to its molar mass.
Thus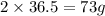 of of hydrochloric acid reacts with 40 g of calcium.
of of hydrochloric acid reacts with 40 g of calcium.
100 g of hydrochloric acid react with=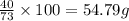 of calcium
of calcium
Thus HCl is the limiting reagent as it limits the formation of product and calcium is the excess reagent as (100-54.79)=45.21 g is left unreacted.
Thus HCl which is a limiting reagent will be used up before Calcium.
Ответ:
The answer is expert opinion
Explanation:
i took the test and got it right