Which of these do not obey the octet rule? select all that apply. select all that apply. clo clo− clo2− clo3− clo4−?
Solved
Show answers
More tips
- F Food and Cooking How to cook crayfish? Everything you need to know...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- C Computers and Internet How to Choose an Uninterruptible Power Supply (UPS) for Your Computer: Expert Tips...
- S Science and Technology How to choose a home theater system?...
- A Auto and Moto How to Choose a Car Wash? Tips and Recommendations...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- S Sport When is the Champions League final?...
Answers on questions: Chemistry
- M Mathematics Determine the range of the graph...
- C Chemistry 1.Chlorine gas (CI2 2.Water(H2,O)3.Hydrogen Bromide (HBr)4.Nitrogen Monoxide(NO)5.Carbon Dioxide (CO2)...
- M Mathematics How do i find the missing value? find the missing value: x^2 = 625 5 10 25 50...
- M Mathematics an oil tank loses 32% of its contents every hour. Peter says the tank will have lost 95% of its original contents by the end of the sixth hour. Is peter correct...
- B Business Carla and john enter into a partnership and form a business. under the partnership agreement, carla is directly responsible for the management of the firm and its...

Ответ:
Further Explanation:
Octet rule: states that for the stability of any element it must have a valence shell of eight electrons (octet means a group of eight). This rule is given by Kossel and Lewis.
Atoms of same or different elements with an incomplete electronic configuration that is having electrons less than 8 are unstable and they combine together to form stable molecules with a complete octet. They can combine by sharing of electrons or loss or gain of electrons.
In , Chlorine (Cl) has 7 electrons in its outer shell and oxygen has 6 electrons in its outer shell. So chlorine and oxygen share 2 electrons each to complete their octet and attain stability. Chlorine atom in the compound has ten electrons in its outer shell so it doesn’t follow the octet rule.
, Chlorine (Cl) has 7 electrons in its outer shell and oxygen has 6 electrons in its outer shell. So chlorine and oxygen share 2 electrons each to complete their octet and attain stability. Chlorine atom in the compound has ten electrons in its outer shell so it doesn’t follow the octet rule.
In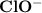 , Chlorine(Cl) has 7 electrons in its outer shell and oxygen has a unit negative charge on it
, Chlorine(Cl) has 7 electrons in its outer shell and oxygen has a unit negative charge on it  so, it has 7 electrons also in its outer shell. So chlorine and oxygen share one electron each to complete their octet and attain stability. All the atoms have eight electrons in their outer shell so they follow the octet rule.
so, it has 7 electrons also in its outer shell. So chlorine and oxygen share one electron each to complete their octet and attain stability. All the atoms have eight electrons in their outer shell so they follow the octet rule.
In , Chlorine (Cl) has 7 electrons in its outer shell and there is 2 oxygen attached to Cl. One has a unit negative charge on it
, Chlorine (Cl) has 7 electrons in its outer shell and there is 2 oxygen attached to Cl. One has a unit negative charge on it  and the other is neutral. So, chlorine and neutral oxygen share 2 electrons each to get a stable octet and form a double bond. Similarly, chlorine shares 1 electron with oxygen ion with a negative charge on it forming a single bond with it. A chlorine atom has ten electrons in its outer shell so the compound doesn’t follow the octet rule.
and the other is neutral. So, chlorine and neutral oxygen share 2 electrons each to get a stable octet and form a double bond. Similarly, chlorine shares 1 electron with oxygen ion with a negative charge on it forming a single bond with it. A chlorine atom has ten electrons in its outer shell so the compound doesn’t follow the octet rule.
In , Chlorine (Cl) has 7 electrons in its outer shell and there is 3 oxygen attached to Cl. One has a unit negative charge on it
, Chlorine (Cl) has 7 electrons in its outer shell and there is 3 oxygen attached to Cl. One has a unit negative charge on it  and the other two are neutral. So, chlorine and both neutral oxygen share 2 electrons to get a stable octet and form a double bond. Similarly, chlorine shares 1 electron with oxygen ion with a negative charge on it, forming a single bond with it. Here, Cl has ten electrons in its outer shell, so it doesn’t follow the octet rule.
and the other two are neutral. So, chlorine and both neutral oxygen share 2 electrons to get a stable octet and form a double bond. Similarly, chlorine shares 1 electron with oxygen ion with a negative charge on it, forming a single bond with it. Here, Cl has ten electrons in its outer shell, so it doesn’t follow the octet rule.
In , Chlorine (Cl) has 7 electrons in its outer shell and there is 4 oxygen attached to Cl. One has a unit negative charge
, Chlorine (Cl) has 7 electrons in its outer shell and there is 4 oxygen attached to Cl. One has a unit negative charge  on it and the other three are neutral. So, chlorine and both neutral oxygen share 2 electrons to get a stable octet and form a double bond. Similarly, chlorine shares 1 electron with oxygen ion with a negative charge on it, forming a single bond with it.
on it and the other three are neutral. So, chlorine and both neutral oxygen share 2 electrons to get a stable octet and form a double bond. Similarly, chlorine shares 1 electron with oxygen ion with a negative charge on it, forming a single bond with it.
Here, Cl has fourteen electrons in its outer shell, so it doesn’t follow the octet rule.
Learn more:
1. The octet rule states that:link
2. How many covalent bond nitrogen forms: link
Answer details:
Grade: Secondary School
Subject: Chemistry
Chapter: Chemical Bonding
Keywords: octet rule, stability, oxygen, chlorine, covalent, sharing, ClO2-, ClO3-, ClO4-, ClO-and ClO.
Ответ:
4.3020 grams of calcium sulfate will be produced.
Explanation:
To calculate the number of moles, we use the formula:
 ....(1)
....(1)
For calcium carbonate:Given mass of calcium carbonate = 3.1660 g
Molar mass of calcium carbonate = 100.0869 g/mol
Putting values in above equation, we get:

For sulfuric acid:Given mass of sulfuric acid = 3.2900 g
Molar mass of sulfuric acid = 98.079 g/mol
Putting values in above equation, we get:
For the given reaction:
By Stoichiometry of the reaction:
1 mole of calcium carbonate reacts with 1 mole of sulfuric acid
So, 0.0316 moles of calcium carbonate will react with =
As, the given amount of sulfuric acid is more than the required amount. Hence, it is present in excess and is considered as an excess reagent.
Calcium carbonate is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:1 mole of calcium carbonate produces 1 mole of calcium sulfate
So, 0.0316 moles of calcium carbonate will produce = of calcium sulfate.
of calcium sulfate.
Now, to calculate the mass of calcium sulfate, we use equation 1:
Molar mass of calcium sulfate = 136.14 g/mol
Moles of calcium sulfate = 0.0316mol
Putting values in equation 1, we get:
Hence, 4.3020 grams of calcium sulfate will be produced.