Fintastichumanoid
01.03.2021 •
Chemistry
Which of these is NOT found in the earths crust?
Oxygen
Iron
Sulphur
Helium
Solved
Show answers
More tips
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Science and Technology Colliders: How They Work and Why They Matter...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- S Social Studies Plz fast which nation has large sectors of both commercial and subsistence farming...
- B Business Initially the market for labor in the California wine industry is in equilibrium, but then the demand for California wines suddenly increases. In the California wine...
- C Chemistry Scientists claim that climate change is happening because Earth is warming. They also claim that humans have helped cause the warming by adding greenhouse gases to the...
- M Mathematics Please help me out! Easy! SOLVE INEQUALITY AND GRAPH ITS SOLUTION! Options above! 1-6(5b+5) 181...
- P Physics Calculate the mass (in si units) of (a) a 160 lb human being; (b) a 1.9 lb cockatoo. calculate the weight (in english units) of (c) a 2300 kg rhinoceros; (d) a 22 g song...

Ответ:
Explanation:
helium
Helium gas is not found in earth crust
Ответ:
28 %
Explanation:
First we have to calculate the moles of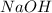
Now we have to calculate the moles of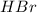
The balanced chemical reaction will be,
From the balanced reaction, we conclude that
As, 1 mole of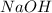 react with 1 mole of
react with 1 mole of 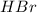
So, 0.019 moles of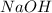 react with=
react with= 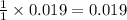 mole of
mole of 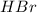
Thus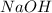 acts as the limiting reagent as it limits the formation of product and [tex}HBr[/tex] is the excess reagent.
acts as the limiting reagent as it limits the formation of product and [tex}HBr[/tex] is the excess reagent.
As, 1 mole of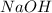 give= 1 mole of
give= 1 mole of 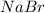
So, 0.019 moles of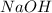 react with=
react with= 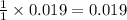 mole of
mole of 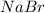
Now we have to calculate the mass of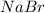
Now we have to calculate the percent yield of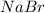
Therefore, the percent yield of sodium bromide is 28%