emthebest123
17.12.2020 •
Chemistry
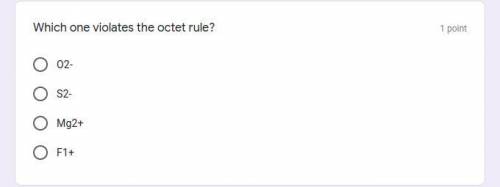
Which one violates the octet rule?
Solved
Show answers
More tips
- L Legal consultation How to Properly Inherit: Tips and Recommendations...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
Answers on questions: Chemistry
- C Chemistry Hey everyone looks like I’m staying I’m am not leaving...
- C Chemistry Matter is cycled throughout the stages of the carbon cycle by...
- C Chemistry Chemical compounds undergo both physical and chemical changes. Which of the following is an example of a physical change?...
- C Chemistry 02.03 quantization of energy lab report...
- C Chemistry (Al = 27.0 g, O = 16.0 g, H = 1.0 g) 2 Al(OH)3 Al2O3 + 3 H2O how many grams are produced from .85 moles of AI(OH)3...
- C Chemistry Design a Cooling Cup for Campers In this task, you’ll design a full-sized cup that’s meant to make drinks cold. The cup you ll design is to be used by campers on extended trips...
- C Chemistry As atomic size increases, how do electronegativity and ionization energy change?...
- C Chemistry Which ones are wrong if the are wrong what is the right answer...
- C Chemistry Which of the following is not a natural toxin that can contaminate food? a. ciguatera b. methyl mercury c. aflatoxins d. dioxins...
- C Chemistry Classify each chemical equation as a combination/synthesis, decomposition, single replacement , double replacement, or combustion a. pb(s) + hg2so4 (s) pbso4(s) +2 hg(i) b. naci(aq)...

Ответ:
F1+ is the one that violates the octet rule
Explanation:
F1+ does not end up having 8 electrons in its valence shell, and there for violates the octet rule, for an atom to comply with the octet rule its charge must reflect the number of electrons it needs to gain or lose to make an octet, but F1+'s charge does not reflect the number of electrons it needs to be stable or have an electron configuration of a noble gas
Ответ:
b
Explanation: