Which represents a balanced nuclear equation?
1) 23/11Na ——>24/11Mg+1/1H
2) 24/11Na ——>24/12Mg+0/-1e
3) 24/13Al ——>24/12Mg+0/-1e
4) 23/12Mg ——>24/12Mg+1/0n
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- F Food and Cooking How to Make the Perfect Glühwein: Step-by-Step Guide...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...

Ответ:
The correct option is 2.
Explanation:
In a nuclear reaction balanced we have that:
1. The sum of the mass number (A) of the reactants (r) is equal to the sum of the mass number of the products (p)
2. The sum of the atomic number (Z) of the reactants is also equal to the sum of the atomic number of the products
So, let's evaluate each option.
1)
The mass number of the reactant is:
The sum of the mass number of the products is:
This is not the correct option because it does not meet the first condition ( ).
).
2)
The mass number of the reactant and the products is:
Now, the atomic number of the reactants and the products are:
This nuclear reaction is balanced since it does meet the two conditions for a balanced nuclear equation, ( and
and 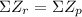 ).
).
3)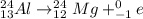
The mass number of the reactant and the products is:
Now, the atomic number of the reactants and the products are:
This reaction does not meet the second condition (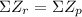 ) so this is not a balanced nuclear equation.
) so this is not a balanced nuclear equation.
4)
The mass number of the reactant and the products is:
This reaction is not a balanced nuclear equation since it does not meet the first condition ( ).
).
Therefore, the correct option is 2.
I hope it helps you!
Ответ:
Nope
Explanation: