Which stable molecule would probably not exist?
a. ch3oh
b. vh2o
c. ch3o
d. c2h2
e. c3h4
Solved
Show answers
More tips
- F Family and Home When and how to start introducing solid foods to your baby?...
- B Business and Finance Moneybookers – What it is and How it Works...
- C Computers and Internet How to Format Your C Drive: Detailed Guide and Tips...
- F Food and Cooking What can and cannot be eaten during Lent?...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- C Computers and Internet What to Do If Your ICQ Gets Hacked?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- S Social Studies Mohandas gandhi s protests during indias independence movement were often successful because of what?...
- E English Read this excerpt from the bachelor s story in The Story-Teller, in which he is describing the character Bertha: Was she pretty? asked the bigger of the small girls. Not...

Ответ:
Option C= CH₃O
Explanation:
CH₃O would not exist as a stable molecules because,
The in this compound oxygen is not stable.
In order to stable the oxygen carbon should form the double bond with oxygen. While the valency of carbon is four. It already form three bonds with three hydrogen atoms in this compound.
There is one electron left to form the bond with oxygen. So, there are not enough electrons which could bonded with oxygen and form stable compound.
Ответ:
Option (d) is the correct answer.
Explanation:
An ion is a specie that contains either a positive or a negative charge.
For example,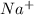 ,
, 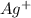 ,
, 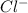 ,
, 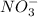 , etc are all ions.
, etc are all ions.
Therefore, an ionic equation is defined as an equation that contains participating dissolved species in the form of ions.
For example,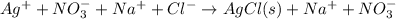
Here, only AgCl is the precipitate as it is insoluble but rest of the species are ions as they have dissolved.
Thus, we can conclude that a complete ionic equation an equation showing all dissolved compounds as ions.