qwemnb7401
28.11.2019 •
Chemistry
Which substance is the limiting reactant when 4.0 g of sulfur reacts with 6.0 g of oxygen and 8.0 g of sodium hydroxide according to the following chemical
2 s(s) + 3 o2(g) + 4 naoh(aq) → 2 na2so4(aq) + 2 h2o(l)
Solved
Show answers
More tips
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- C Chemistry Limited ATP Synthesis in the Dark in a laboratory experiment, spinach chloroplasts are illuminated in the absence of ADP and Pi, then the light is turned off and ADP and Pi are added....
- C Chemistry Calculate the percent error in the atomic weight if the mass of a Zn electrode increased by 0.3681g and 6.514x10-3 moles of Zn was produced. You may assume the molar mass of elemental...
- C Chemistry Nickel (III) cyanide + aluminum permanganate Produces?...
- C Chemistry How many grams of Mg(NO3), are necessary to make 1500 mL of a 0.50 M solution?...
- C Chemistry How many grams of carbon dioxide are produced when 255 grams of octane (C3H18) combust?...
- C Chemistry What is the rate law for step 2 of this reaction? Express your answer in standard MasteringChemistry notation. For example, if the rate law is k[A][C]3 type k*[A]*[C]^3....
- C Chemistry As a rule of thumb, if the [ H3O ] is less than 1% of the value of the [Acid], it does not have to be considered when figuring the concentration of the acid. Use your results in...
- C Chemistry Radioactive elements decay at a know rate known as half-life. Choose all of the correct statements concerning half-life. Carbon-14 has a half-life of 5,730 years. A 30 gram sample...
- C Chemistry A mystery atom has 17 protons, 17 electrons, and 18 neutrons. Which element is it...
- C Chemistry An element is dull, is poor conductor of heat and electricity and needs one electron to complete its valence shell (outermost energy level). This element most likely belongs to which...

Ответ:
Limiting reactant is NaOH
Explanation:
In the reaction:
2S(s) + 3O₂(g) + 4NaOH(aq) → 2Na₂SO₄(aq) + 2H₂O(l)
4.0 g of sulfur, 6.0 g of oxygen and 8.0 g of sodium hydroxide are:
4,0g S×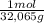 = 0,125 moles S
= 0,125 moles S
6,0g O₂× = 0,188 moles O₂
= 0,188 moles O₂
8,0g NaOH× = 0,2 moles NaOH
= 0,2 moles NaOH
For a complete reaction of 0,2 moles of NaOH you will need:
0,2 mol NaOH× = 0,1 moles of Sulfur
= 0,1 moles of Sulfur
0,2 mol NaOH× = 0,15 moles of Oxygen
= 0,15 moles of Oxygen
As you have 0,125 moles of S and 0,188 moles of O₂ that are more than moles you need for a complete reaction of NaOH, limiting reactant is NaOH
I hope it helps!
Ответ:
polystyrene
Explanation:
it weighs less and is able to float on water, an aluminum wood slate would sink in water