Why does a salt compound give off light (or a colored flame) when burned? 4. did lithium chloride and sodium chloride give off similar colors? why or why not? 5. explain what you think causes different compounds to give off different flame colors. 6. using your results, identify which substances you would put in a firework to delight a crowd of spectators?
Solved
Show answers
More tips
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
- A Animals and plants How to Teach Your Parrot to Talk?...
Answers on questions: Chemistry
- C Chemistry Element compound mixture...
- B Biology Science test 1. how do some hormones affect only certain cells in the body but not others? only cells having specific receptors for a hormone can respond to the hormone...
- W World Languages What s the best definition of a topic sentence? a. the first sentence of the paragraph b. the statement in a paragraph that provides the central idea c. the conclusion...
- B Biology Need help ASAP. 1.) Explain the difference or similarities between CRISPR and at least one other type of Genetic Engineering Technique you have researched? 2.) What...
- S SAT Where on the physical activity pyramid do sedentary activities belong?the physical activity pyramid: top level of pyramid is sedentary activities, second level is anaerobic...
- G Geography Which three empires affected ancient india? egyptian persian roman chinese greek huns...

Ответ:
The salt compounds give off light due to their emission spectrum.
(4) , lithium chloride and sodium chloride do not give off similar colors because of the difference in their emission spectrum.
, lithium chloride and sodium chloride do not give off similar colors because of the difference in their emission spectrum.
(5) Flame colors are characteristics of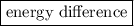 between the two levels, which is different for different elements.
between the two levels, which is different for different elements.
(6) can be put in a firework.
can be put in a firework.
Further explanation:
Electronic transition:
An electronic transition is a process that occurs when an electron undergoes emission or absorption from one energy level to another energy level.
When an electron undergoes a transition from lower energy level to the higher energy level then it requires energy to complete the process. This transition is an absorption process.
When an electron undergoes a transition from higher energy level to lower energy level then it emits energy to complete the process. This transition is an emission process.
The formula to calculate the difference between two energy levels of an atom is,
Where,
The salt compound gives off the light because its electrons absorb energy and get excited to some higher energy level. But when it returns to the lower level, it emits energy in the form of light, resulting in emission spectrum.
(4) No, lithium chloride and sodium chloride do not give off similar colors. This is because both these compounds have different allowed energy levels for their respective electrons. Lithium ion shows a red emission spectrum while sodium ion shows yellow emission spectrum.
(5) Different elements have different allowed energy levels for electrons. When an atom is heated, it absorbs energy and reaches some higher energy level. When an electron jumps from higher energy level to lower energy level, it emits energy in form of light. The wavelength of light depends on the energy difference between the two levels, which is different for different elements, and therefore different elements show different flame colors.
(6) Salts of alkali and alkaline earth metals can be used in fireworks as these are highly electropositive metals and give off electrons so easily and bright colored flames are produced. For example, barium chloride gives off the green flame when burned.
Learn more:
1. Component of John Dalton’s atomic theory:
2. Basis of investigation for the scientists:
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Atomic structure
Keywords: electronic transition, barium chloride, colored flames, alkali, alkaline earth metals, electropositive, emission, absorption, electrons, allowed energy levels, higher level, lower level.
Ответ:
Thalia would need to consider that the sculpture's inertia is keeping the object in place. Newton's first law applies to this situation. The inertia would have to be overcome to enable the sculpture to move, but the inertia would also keep the sculpture moving after its movement was started. The force that is required to move the sculpture would depend on the mass and acceleration at which Thalia wanted the sculpture to move. Finally, any force applied to the sculpture would be resisted by the sculpture in an equal and opposite manner.