rachelsweeney10
07.07.2019 •
Chemistry
Would it take 5 cell divisions for one original cell to produce 128 new cells?
Solved
Show answers
More tips
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
Answers on questions: Chemistry
- C Chemistry How many hydrogen atoms are in 5.20 mol of ammonium sulfide?...
- C Chemistry The chemical equation for the reaction of baking soda (sodium bicarbonate, nahco3) and vinegar (acetic acid, ch3cooh) may be written as two steps. fill in the missing information...
- C Chemistry Abeaker of nitric acid is neutralized with calcium hydroxide. write a balanced molecular equation for this reaction...
- C Chemistry From the data below, calculate the total heat (in j) needed to convert 0.782 mol of gaseous ethanol at 300.0°c and 1 atm to liquid ethanol at 25.0°c and 1 atm...
- C Chemistry (i) The element Francium is extremely rare and very little is known about its chemical and physical properties Use the following data to estimate its density and ionization energy.Density...
- C Chemistry Name and explain all the types of Energy and provide an example for each....
- C Chemistry Hola quien me ayuda doy corona :3...
- C Chemistry What kind of weather does a cold front usually bring? Please select the best answer from the choices provided A warm B stormy C sunny D windy...
- C Chemistry The standard heat of formation for sulfur dioxide gas is 296.8 kJ/mol. Calculate the amount of energy given off when 34 g of SO2(g) is formed from its elements. Answer in units...
- C Chemistry CS2, Cl2, HF, HCI, H20, H2S which of the following have a dipole moment...

Ответ:
Yes, it will take 5 cell divisions for one original cell to produce 128 cells.
Ответ:
Explanation: Here, we will be considering 1 cup is equal to 1 mol.
2 water + sugar + lemon juice → 4 lemonade
Moles of water present in 946.36 g of water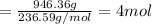
Moles of sugar present in 196.86 g of water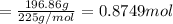
Moles of lemon juice present in 193.37 g of water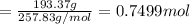
Moles of lemonade in 2050.25 g of water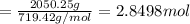
As we can see that number of moles of lemon juice are limited.
So, we will consider the reaction will complete in accordance with moles of lemon juice.
1 mole lemon juice reacts with 2 mol of water,then 0.7499 mol of lemon juice will react with:
Mass of water used = 1.4998 mol × 236.59 g/mol=354.8376 g
Water remained unused = 946.36 g - 354.8376 g =591.5223 g
1 mole lemon juice reacts with mol of sugar,then 0.7499 mol of lemon juice will react with:
Mass of sugar used = 0.7499 mol × 225 g/mol = 168.7275 g
Sugar remained unused = 196.86 g - 28.1325 g
1 mole of lemon juice gives 4 moles of lemonade.
Then 0.7499 mol of lemon juice will give:
Mass of lemonade obtained = 2.996 mol × 719.42 g/mol = 2157.9722 g
Theoretical yield of lemonade = 2157.9722 g
Experimental yield of lemonade = 2050.25 g
Percentage yield of lemonade:
The percentage yield of lemonade is 95% and ingredient which remained unused were water and sugar.