davisnaziyahovz5sk
30.10.2021 •
Chemistry
Write the uses of Ions .
Solved
Show answers
More tips
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry 5. a. How many mol of sodium chloride, NaCl, are required to prepare 0.2 L of a 3 M solution? b. How many grams is this (hint: molar mass of NaCl = U?...
- C Chemistry What has the shortest or smallest wavelength....
- C Chemistry A student is doing experiments with CO2(g). Originally a sample of the gas is in a rigid container at 299k...
- C Chemistry 5 Select the correct answer. Object A with a mass of 500 kilograms hits stationary object B with a mass of 920 kilograms. If the collision is elastic, what happens...
- C Chemistry Which of the following is not a reason to question the information one receives? a. It agrees with previous experience. b. It does not agree with previous experience....
- C Chemistry Is the longest stage of the cell cycle....
- C Chemistry In terms of bonds, what would the molecule c₃h₆ be classified as? inorganic compound alkene alkyne alkane...
- C Chemistry What would a general formula of an acid always start with? what does the general formula of an acid have to end with? where would you put a subscript in a general...
- S Spanish How do you spell 20, 21, 13, in Spanish or say...
- M Mathematics What is the length of a marathon in (1mile=1.6kilometers) ?...

Ответ:
This is what they are: An ion is a charged atom or molecule. It is charged because the number of electrons do not equal the number of protons in the atom or molecule. An atom can acquire a positive charge or a negative charge depending on whether the number of electrons in an atom is greater or less then the number of protons in the atom.
This is their purpose: Once they reach our bloodstream, negative ions are believed to produce biochemical reactions that increase levels of the mood chemical serotonin, helping to alleviate depression, relieve stress, and boost our daytime energy.
Explanation:
Ответ:
Explanation:
Given parameters:
Mass of the sample = 0.622g
Volume of the gas = 2.4L
Temperature = 287K
Pressure of the gas = 0.85atm
Unknown
Molar mass of the gas = ?
Solution
To find the molar mass of the gas, we first assume that the gas in question is an ideal gas i.e. it obeys Boyle's and Charles's law.
On this premise, we can find the unknown which is the molar mass of the gas.
We first use the ideal gas equation to find the number of moles of the gas since all the parameters apart from the number of moles are given:Solving for number of moles:
The ideal gas equation is given as PV = nRT
We make the unknown "n" the subject of the equation
n =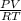
R is the gas constant whose value is 0.082atmdm³mol⁻¹K⁻¹
n =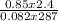
n =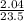 = 0.087mole
= 0.087mole
Now, we know the number of moles of the gas. We can proceed to find the molar mass from the given mass and the number of moles calculated using the equation below:molar mass =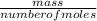
molar mass =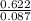 =7.15gmol⁻¹
=7.15gmol⁻¹
Part 2:
If the sample was placed under a low temperature, we want to consider what would happen to the volume:
Since we assume that the gas is an ideal gas, we can explain what will happen to the gas using Charles's law. The law states that "The volume of a fixed mass of a gas varies directly as its absolute temperature if the pressure is constant".
If we assume the pressure is constant for this gas, the volume of the gas would decrease as the temperature becomes lower. We would get lower values for both actual volume and predicted volume.