You fill a tank with gas at 30°c and 101.5 kpa and seal it. you decrease the temperature to 10°c but keep the volume constant. what happens to the pressure? (careful!
Solved
Show answers
More tips
- H Health and Medicine Choosing the Right Contraception Method: Your Ultimate Guide...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- B Biology Mitosis is the type of cell division that ___ select multiple answers!!can cause cancer if not controlledgenerates gametes for sexual reproductioncuts down the number of chromosomes...
- E English The great gatsby chapter 2 compare/contrast the personalities and physical characteristics of myrtle and daisy....

Ответ:
PV = nRT
When V, n, and R are all constant, we can say that,
(T1)(P2) = (T2)(P1)
We convert temperature into kelvin (This is the portion where we have to be very careful not using the degrees Celsius).
(30 + 273)(P2) = (10 + 273)(101.5 kPa)
The value of P2 is 94.8 kPa.
Ответ:
The correct answer is Option A.
Explanation:
All the radioactive decay processes follows first order kinetics.
To calculate the rate constant for a reaction, we use the equation:
where,
k = rate constant for a reaction
Putting all the values in above equation, we get:
To calculate the amount of sample left, we use the equation:
where,
N = amount of sample left after time 't'
k = rate constant of the reaction =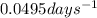
t = time taken = 56 days
Putting values in above equation, we get:
Hence, the correct answer is Option A.