andersonjalen34
23.11.2021 •
Chemistry
You measured electrochemical cell potentials with a Cu2+/Cu couple as the anode. Predict the results you would observe if Ag+/Ag were the anode.
half-cell predicted potential
Cu2+/Cu difference in V:
Zn2+/Zn -0.46
?
Solved
Show answers
More tips
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
Answers on questions: Chemistry
- B Business Jacob and molly decide to start a new cake-decorating business. they each contribute $10,000 to get the business off the ground. what kind of sources this money is considered?...
- H History Ch of the followingish written document that lays out the structure of the us government...
- M Mathematics Which numbers are solutions to the inequality-3x-3 6...

Ответ:
The given question is incomplete. The complete question is as follows.
Calculate the pressures of NO, , and NOCl in an equilibrium mixture produced by the reaction of a starting mixture with 8.2 atm NO and 4.1 atm
, and NOCl in an equilibrium mixture produced by the reaction of a starting mixture with 8.2 atm NO and 4.1 atm  . (Hint: KP is small; assume the reverse reaction goes to completion then comes back to equilibrium.)
. (Hint: KP is small; assume the reverse reaction goes to completion then comes back to equilibrium.)
Explanation:
According to the ICE table,
Initial: 8.2 4.1 0
Change: -4.1x -x +4.1x
Equilbm: (8.2 - 4.1x) (4.1 - x) +4.1x
Now, expression for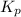 of the reaction is as follows.
of the reaction is as follows.
x = 1.9
Therefore, at equilibrium
[NOCl] =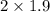 = 3.8
= 3.8
[NO] = (8.2 - 7.79) = 0.41