NorbxrtThaG
21.04.2020 •
Engineering
Using EES (or other) software, calculate and plot the entropy change of air across the normal shock for upstream Mach numbers between 0.5 and 1.5 in increments of 0.1. Explain why normal shock waves can occur only for upstream Mach numbers greater than Ma 5 1.
Solved
Show answers
More tips
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Engineering
- M Mathematics What is 4.110 in word form...
- M Mathematics What is 3 9/100 in decimal...
- E English Write 15,409 in two other forms...
- E English Sentence using direct democracy...
- C Chemistry Alcohols always contain groups....
- M Mathematics What is the midpoint of 9.9 of 10...
- M Mathematics What percent of 60 is 96...
- M Mathematics Suppose f(x)=. Find the graph of f(). Graph 1 Graph 2 Graph 3 Graph 4...
- M Mathematics Bulunan hazine sandığı üzerinde kilidin şifresi iki asal sayının çarpımıdır yazmaktadır A) 189B)171C)126D)123 ...
- H History Discuss the Pluralist...

Ответ:
Reaction A has a higher net reaction rate
Explanation:
Rate of electrochemical reaction rate per unit area =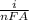
F = 96500 C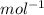 ( Faraday constant)
( Faraday constant)
n = number of moles of electrons per mole of reactant
A = Area of electrode
i = current generated
convert the current from Amperes to C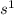
To calculate net reaction of reaction A
i = 5 c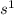
n = 2
A = 2
back to the equation
= 5 / (2 * 96500 * 2) = (1.3 * 10 ^ -5) mols^-1 cm^-2
To calculate net reaction of reaction B
i = 15 c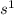
n = 3
A = 5
back to the equation
= 15 / ( 3 * 96500 * 5) = (1.036 * 10 ^ -5) mols^-1 cm^-2