All of the following received veto power in the UN Security Council EXCEPT
France
Us
USSR
Japan
China
Solved
Show answers
More tips
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: History
- H History Which of the following statements concerning the amazon region of brazil is true? a large areas of the rain forest are being destroyed. b brazil’s government has forbidden logging,...
- B Business A random sample of records of sales of homes from February 15 to April 30, 1993, from the files maintained by the Albuquerque Board of Realtors gives the Price and Size (in square...
- E English What is Mrs. Baroda s impression of Gouvernail after meeting him for the first time? a. She likes him for some reason she can t explain. b. She does not like him because he s...
- C Chemistry Observe this chemical reaction: 4 Fe + 302- 2Fe 203 How many atoms of oxygen are in this !...

Ответ:
Japan.
Explanation:
The United Nations Security Council "veto power" refers to the power of the five permanent members of the UN Security Council (China, France, Russia, the United Kingdom and the United States) to veto any "substantive" resolution.
Ответ:
The unit cell edge length for the alloy is 0.405 nm
Explanation:
Given;
concentration of Ag,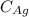 = 78 wt%
= 78 wt%
concentration of Pd, = 22 wt%
= 22 wt%
density of Ag = 10.49 g/cm³
density of Pd = 12.02 g/cm³
atomic weight of Ag,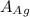 = 107.87 g/mol
= 107.87 g/mol
atomic weight of iron,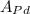 = 106.4 g/mol
= 106.4 g/mol
Step 1: determine the average density of the alloy
Step 2: determine the average atomic weight of the alloy
Step 3: determine unit cell volume
for a FCC crystal structure, there are 4 atoms per unit cell; n = 4
Step 4: determine the unit cell edge length
Vc = a³
Therefore, the unit cell edge length for the alloy is 0.405 nm