jimmyjimjim
16.02.2021 •
Mathematics
How about more free points
Solved
Show answers
More tips
- F Family and Home When and how to start introducing solid foods to your baby?...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- F Food and Cooking Homemade French Fries: The Ultimate Guide...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Mathematics
- M Mathematics 2,12,8 = 156, 14,4= 353,63,7= XFIND X...
- M Mathematics Given that tan theta=-1 what is the value of sec theta for 3pi/2 a. - squareroot b. Squareroot c. 0 d. 1...
- B Biology Blank can disrupt cellular homeostasis and can even lead to uncontrolled cell division and the formation of cancers. (and it is not carcinogens because i got that wrong)...
- B Biology Which of the following was used to measure the rate of photosynthesis?...
- S Social Studies Which kind of case automatically comes before Georgia s supreme court?...
- S Social Studies What problems can you imagine arising from a nation of mixed nationalities and faiths? provide real world examples, if you can. plz try, any answers will do....

Ответ:
yay
Step-by-step explanation:
thanks
Ответ:
thank u
Step-by-step explanation:
Ответ:
At t = 2 minutes, remaining quantity of the radioactive element is 12.5 mg.
Step-by-step explanation:
To get the answer of this question we will solve this further with the help of the equation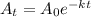
where k = decay constant
t = time for decay
From the graph attached we can say that 50 mg of a radioactive element remained half in 1 minute.
So the equation becomes
Now we take natural log on both the sides of the equation
ln50 = ln[25.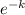
3.912 = ln25 +
3.912 = 3.219 + (-k)lne
3.912 - 3.219 = -k [since lne = 1]
0.693 = -k
k = -0.693
Now we will calculate the remaining quantity of the element after 2 minutes
=
=
=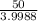
= 12.50 mg
Now we confirm this value from the graph.
At t = 2 minutes, remaining quantity of the radioactive element is 12.5 mg.