KhaliylTestman
16.07.2020 •
Mathematics
How do I solve this problem?
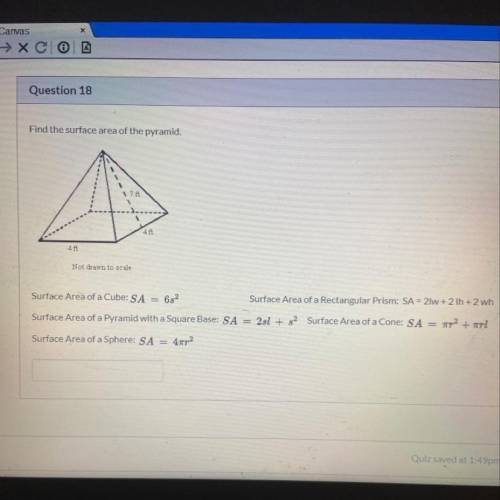
Solved
Show answers
More tips
- H Health and Medicine Impeccable Memory: How to Improve It...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport When will the Biathlon World Championships 2011 take place in Khanty-Mansiysk? Answers to frequently asked questions...
- H Health and Medicine Trading Semen for Money: Where Can You Sell and Why Would You Want to?...
- F Food and Cooking Homemade French Fries: The Ultimate Guide...
- H Health and Medicine How to Increase Blood Pressure without Medication?...
- S Style and Beauty Choosing a Hair Straightener: Specific Criteria to Consider...
- F Food and Cooking How to Make Polendwitsa at Home?...
- S Science and Technology When do we change our clocks?...
- L Leisure and Entertainment What to Give a Girl on March 8?...
Answers on questions: Mathematics
- M Mathematics Angles and arcs can be measured in degreased or in ?...
- M Mathematics Spiral Review Calculate the slope of the line that passes through (3, 2) and (-7,4). Enter the correct answer in the box....
- M Mathematics Tell whether or not the measurements below form a triangle: 1. 4, 5, 10 2. 12, 26, 20 3. 2, 3, 5...
- M Mathematics I need help on this im struggling...
- M Mathematics Is is square root of 1.6875 a rational number...
- M Mathematics Please Hurry Find the value of x...
- E English I Need Help On This, I ll Mark Whoever Answers First Brainliest!...
- H History Consider the goods traded in these six cities. would you say they were luxury items for the wealthy or everyday items for the common people? give example...

Ответ:
Ответ:
45 g de oxido de hierro (III) liberarán 18.9 L de CO₂
Step-by-step explanation:
La reaccion quimica es la siguiente:
Fe₂O₃ (s) + 3CO (g) → 2Fe (s) + 3CO₂ (g)
1 mol de oxido de hierro (III), reacciona con 3 moles de monoxido de carbono para liberar 3 moles de CO₂ y 2 moles de Fe sólido.
Pasamos la masa de F₂O₃ a moles:
159.6 g . 1 mol /159.7g = 1 mol de oxido
Entonces, aplicamos la regla de tres:
1 mol de oxido produce 3 moles de dioxido de carbono, indudablemente eso será lo que formemos de producto, por lo tanto 3 moles de CO₂ estan ocupando 67.2 litros.
Ahora calculamos los nuevos moles:
45g . 1 mol /159.7 g = 0.282 moles
Aplicamos la regla de tres:
Si un mol de oxido produce 3 moles de CO₂
0.282 moles producen (0.82 . 3 ) = 0.845 moles de CO₂
Como tenemos menos moles de gas, el volumen ocupado sera mayor, hacemos la regla de tres:
Si 3 moles de CO₂ ocupan 67.2 litros
0.845 moles ocuparan (0.845 . 67.2/ 3 ) = 18.9 litros