TheOriginal2x
30.03.2020 •
Mathematics
How many grams of NH3 are required to produce 4.65 g of HF
Solved
Show answers
More tips
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- C Computers and Internet 3D Glasses! What is this thing?...
- C Computers and Internet How to Reinstall Windows: A Detailed Guide for Beginners...
Answers on questions: Mathematics
- M Mathematics What is 224 blocks with 8 blocks in each row how many rows would i make...
- M Mathematics Mr Keen, a band teacher wanted to know if certain types of instruments are more appealing to one gender or the other. So he conducted a survey of his students preferences....
- M Mathematics What weight is shown ?...
- S Social Studies During Harry Truman s presidency, food and supplies were dropped to the people of Berlin, Germany. They were experiencing starvation because of blocks set up by the...
- M Mathematics To say that a coin is fair means that when it is tossed, each of the outcomes (heads or tails) has a 50% probability of occurring. This means: a. in the next six flips...
- S Spanish How do you say stay in your lane in spanish? use it in a sentence : )...

Ответ:
1.326 grams of NH3 are required to produce 4.65 g of HF.
Step-by-step explanation:
Balanced chemical reaction is written first to know the number of moles taking part in original reaction.
NH3 +3 F2 ⇒3 HF + NF3
Given:
mass of HF = 4.65
First the number of moles of HF in 4.65 grams is calculated by using the formula:
number of moles (n) =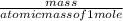
atomic mass of HF = 20 grams/mole
putting the values in the above equation number of moles can be found.
n =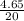
= 0.235 moles of HF are given.
From the equation it can be said that:
1 mole of NH3 reacts to form 3 moles of HF
so, x moles of NH3 would react to form 0.235 moles of HF
3x = 0.235
x =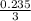
x = 0.078 moles of NH3 is required.
The moles are converted to mass by applying the formula:
mass = atomic mass X number of moles (atomic mass of NH3 = 17 grams/mole)
putting the values in the formula
mass = 17 X 0.078
mass = 1.326 grams
Ответ:
There are a few ways to find out the correct answer. One of them is to divide 2.40 by 6 to get a price of one cake and then multiply it by 10:
2.40 ÷ 6 = 0.40
0.40 * 10 = 4.00
Ten cakes would cost 4 pounds.