shaylakabler333
09.12.2020 •
Mathematics
Someone help me with this aswell

Solved
Show answers
More tips
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Mathematics
- M Mathematics 8x-4(x+8)=8. what s the answer, i need the work as well...
- M Mathematics .How much force is needed to accelerate a truck with a mass of 2,000 kg, at a rate of 3 m/s²?...
- M Mathematics Maya works in a high-end consignment clothing store and earns a commission of 4% on all of her sales. If she sells $1500 worth of clothing, what is her commission?...
- M Mathematics Given the similar triangles. Find the missing side....
- M Mathematics Solve my problems please , now now!...
- M Mathematics Find m2ABC if m2ABC = 4x + 9 and m2 EBD = 7x– 9. A6 B 33 C 45 D 73...
- H History 2. Around 2050 BCE a noble named defeated his rivals and united Egypt....

Ответ:
jerry is right because s represents how many pairs of shoes are there but the question is to find how many shoes are there so
jerry multiplied the number of pairs(s) × 2
so he got 2s and hence jerry is right
Ответ:
The sample is 13414 years old.
Step-by-step explanation:
Expression for rate law for first order kinetics is given by:
where,
k = rate constant
t = age of sample
a = let initial amount of the reactant= 100
a - x = amount left after decay process=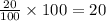
a) for completion of half life:
Half life is the amount of time taken by a radioactive material to decay to half of its original value.
b) for completion of 20% of reaction
The sample is 13414 years old.