uc22912
12.02.2020 •
Mathematics
Two containers, X and Y, are each filled by an ideal gas at the same temperature. The volume of Y is half the volume of X. The number of moles of gas in Y is three times the number of moles of the gas in X. The pressure of the gas in X is PX and the pressure of the gas in Y is PY. What is the ratio LaTeX: \frac{P_X}{P_Y}P Y P Y
Solved
Show answers
More tips
- W Work and Career What is the Most In-Demand Profession in the Modern World?...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Mathematics
- M Mathematics 3a+4=0 What’s the solution...
- M Mathematics Andrew has a salary of £250. he shares his salary with his sister in the ratio 2: 3 every week. he saves 12% each week. how many weeks will it take for him to save £360?...
- M Mathematics Complete factorization of 6x^2?...
- B Biology (please help me) Select the correct answer.Mendel performed studies on the seed color of pea plants. What conclusion can be drawn from this diagram?...
- P Physics A193nm-wavelength uv laser for eye surgery emits a 0.500mj pulse. (a) how many photons does the light pulse contain?...
- B Biology Areceptor that monitors stretch of the patellar tendon can be classified as a) mechanoreceptor and interoceptor. b) mechanoreceptor and proprioceptor. c) nociceptor and exteroceptor....

Ответ:
The answer to the question is
The ratio of the two gas pressures , that is Px to Py = 1/6
, that is Px to Py = 1/6
Step-by-step explanation:
Let the gases Volumes be V₁ and V₂
Where volume of X = V₁ and
volume of Y = V₂
The volume of Y is half the volume of X
∴ V₂ =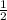 × V₁
× V₁
Let the number of moles be n₁ and n₂ in X and Y respectively
therefore n₂ = 3 × n₁
The pressure of the gas in X is Pₓ and the pressure of the gas in Y is then we have
then we have
P₁ × V₁ = n₁ × R × T₁ , and P₂ × V₂ = n₂ × R × T₂
(P₁ × V₁)/(n₁ × T₁) = (P₂ × V₂)/(n₂ × T₂)
but T₁ = T₂
Therefore
(P₁ × V₁)/n₁ = (P₂ × V₂)/n₂. However n₂ = 3 × n₁ and V₂ =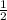 × V₁ therefore substituting in the equation we have
× V₁ therefore substituting in the equation we have
(P₁ × V₁)/n₁ = (P₂ ×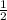 × V₁ )/(3 × n₁) from where
× V₁ )/(3 × n₁) from where
P₁ /P₂ = (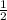 × V₁ × n₁)/(V₁×3 × n₁) =0.5/3 = 1/6
× V₁ × n₁)/(V₁×3 × n₁) =0.5/3 = 1/6
The ratio of = 1/6
= 1/6
Ответ:
Let x be the mysterious number. So...
2x + 3 = 20
Subtract both sides by 3:
2x = 17
Divide both sides by 2:
x = 17/2