Acarnot engine whose low-temperature reservoir is at 19.4°c has an efficiency of 29.7%. by how much should the celsius temperature of the high-temperature reservoir be increased to increase the efficiency to 62.3%?
Solved
Show answers
More tips
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine 10 Simple and Effective Tips on How to Lose Weight in a Week...
- F Food and Cooking What s the Best Rice for Cooking Plov?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics Secrets of NATO: Which Countries are Part of the Alliance?...
Answers on questions: Physics
- P Physics Sketch a velocity grapn that has a negative acceleration and a negative initial velocity...
- P Physics A commutator is used to continually reverse the current flow to maintain rotation of the loop. Select one: O True False...
- P Physics Calculate the work done....
- P Physics Why the impact of the loaded truck on a body coming it’s the way is very large even if the truck is moving slowly...
- P Physics For every 100 joules of useful energy output , the heater wastes 100 joules , find the input energy of the heater....
- P Physics The horizontal surface on which the three blocks with masses M, 2M, and 2M slide is frictionless. The tension in string 1 is T1 = 12 N. Find F....
- B Biology 15. Explain the difference between the law of conservation of energy and conserving energy. How can conserving energy help prevent energy shortages?...
- S Social Studies Sudhir Venkatesh, the author of the book, mentions that the field of sociology has long been divided into two camps in terms of methods of doing research. Understand the two...
- M Mathematics Uppose that the dollar cost of producing x appliances is c(x)equals900plus80xminus0.2xsquared. a. Find the average cost per appliance of producing the first 150 appliances....
- M Mathematics A population of y coyotes in a national park triples every year for 20 years. The function y = 13(3)* represents the population, where x is the number of 20-year periods....

Ответ:
The Temperature of the Hot reservoir should be increased by 359.86°C
Explanation:
A Carnot engine is a reversible engine, that means that it can work both as a heat engine and as a refrigerator, it can both use heat from a hot reservoir (part of which later must end up in a cold reservoir) to produce work, or use work to move heat from a cod to a hot reservoir.
The efficiency of a carnot engine working as a heat engine is given by:
were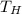 and
and 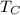 are the Absolute temperatures of the hot and cold reservoir respectively.
are the Absolute temperatures of the hot and cold reservoir respectively.
To get the absolute temperature in K from the relative temperature in °C, we must add 273.15 K (the absolute temperature of the freezing point of water at atmospheric pressure)
Thus:
Now, if we know the Temperature of the cold reservoir, and the engine's efficiency, we can use that information to get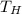 as follows:
as follows:
Now, that is the Temperature of the hot reservoir to begin with, but if we want a higher efficency, we need to increase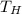 until
until 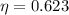 .
.
To find out what value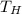 has to reach, we repeat the same calculation as before, except now
has to reach, we repeat the same calculation as before, except now 
So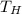 has increased from
has increased from 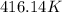 to
to 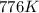 , that means an increase in temperature of :
, that means an increase in temperature of :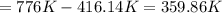
This increment of 359.86K is also an increment of 359.86°C, because increments are relative measures of temperature.
Ответ:
359.67°C or 359 K
Explanation:
Case I:
T2 = 19.4°C = 19.4 + 273 = 292.4 K
η = 29.7 % = 0.297
Let T1 be the temperature of hot reservoir.
By using the formula for the efficiency of Carnot's heat engine
T1 = 415.93 k
Case II:
T2 = 19.4°C = 19.4 + 273 = 292.4 K
η = 62.3 % = 0.623
Let T'1 be the temperature of hot reservoir.
By using the formula for the efficiency of Carnot's heat engine
T'1 = 775.6 k
Incraese in the temperature of hot reservoir
= T'1 - T1 = 775.6 - 415.93 = 359.67°C or 359 k
Ответ: