Amass of 1.4 kg of water at 22 degrees celcius is poured from a height of 2.5 m into a vessel containing 5.0 kg of water at 22 degrees celcius. how much does the internal enetgy of the 6.4 kg of water increase? is it likely that the water temperature increases? explain
Solved
Show answers
More tips
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...

Ответ:
Explanation:
Given
mass of water at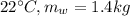
Height of water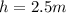
Internal Energy is defined as Energy Posses by all the molecules except macroscopic Kinetic and Potential Energy
So Potential Energy of water is used to increase the internal Energy
Potential Energy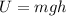
this Energy increase the internal Energy
Since Internal Energy is a function of Temperature so with increase in internal Energy will cause the temperature to rise.
Ответ:
Object 1 had faster-moving particles