krazziekidd2p845ri
27.08.2020 •
Physics
As you did above for the gas, now estimate the number of molecules found in a cubic centimeter of a liquid at ordinary pressure and temperature.
Solved
Show answers
More tips
- F Food and Cooking What to Give as a Wedding Gift?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
Answers on questions: Physics
- M Mathematics NEED HELP ASAPThis scatter plot shows the relationship between the amount of time, in hours, spent working on an essay and the grade, as a percent, earned. Enter the approximate...
- P Physics acontainer is filled whith mercury to alevel of 10m whit water to alevel of 8m and whit oil to alevel of 5m the densities oil ,water and mercury are 0.8g/cm³,1g/cm³ and 13.6g/cm³...
- P Physics The path of the earth around the sun is an ellipse with the sun at one focus. The ellipse has a major axis of 186,000,000 miles and eccentricity of 0.017. Find the distance between...
- M Mathematics An employee currently makes $10 per hour and will be given yearly raises if performance evaluations are satisfactory. assuming annual evaluations are satisfactory, which statement...

Ответ:
There are molecules in a cubic centimeter of water at ordinary pressure and temperature.
molecules in a cubic centimeter of water at ordinary pressure and temperature.
Explanation:
Let suppose that liquid is water at a pressure of a atmosphere and a temperature of 25 ºC. Due to incompresibility of liquids, water density does not have any change of importance due to changes in pressure and temperature.
Density and molar mass of water are 1 gram per cubic centimeter and 18.015 grams per mole. The mass of water in a cubic centimeter (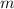 ), measured in grams, is:
), measured in grams, is:
Where:
Given that and
and  , the mass of water is:
, the mass of water is:
The amount of moles ( ) inside the sample is:
) inside the sample is:
Where:
If and
and  , then:
, then:
According to the Avogadro's Principle, there are molecules per mole. Hence, the number of molecules in a cubic centimeter of water at ordinary pressure and temperature is determined by simple rule of three:
molecules per mole. Hence, the number of molecules in a cubic centimeter of water at ordinary pressure and temperature is determined by simple rule of three:
There are molecules in a cubic centimeter of water at ordinary pressure and temperature.
molecules in a cubic centimeter of water at ordinary pressure and temperature.
Ответ:
You can make an answer brainliest by selecting a yellow button above an answer that says mark as brainlist or something along those lines