lolaloiuy7695
02.08.2019 •
Physics
Asingle covalent bond forms when two atoms do which of these?
Solved
Show answers
More tips
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
Answers on questions: Physics
- M Mathematics If point (-1,0) is on the line whose equation is y=2x+b, what is the value of b?...
- M Mathematics Graph the system of linear inequalities. Your graph should demonstrate that you re following the instructions below. Use x- and y- intercepts to graph inequality A. Use...
- M Mathematics The number of days between aug. 9 and jan. 3 is...
- B Biology The third layer of the eye, the receives light rays and images and passes them on to the __ nerve. sclera choroid retina, auditory olfactory optic...

Ответ:
Ответ:
A single covalent bond is formed when two atoms share a pair of electrons.
Explanation:
Valencia electrons are the electrons found in the last electronic layer (called valence orbitals). These electrons are what determine the ability of the atom to form bonds. When an element joins another, it does so through its valence electrons.
The covalent bond is the chemical bond between atoms where electrons are shared, forming a molecule. Covalent bonds are established between non-metallic elements, such as hydrogen H, oxygen O and chlorine Cl. These elements have many electrons in their outermost level (valence electrons) and have a tendency to gain electrons to acquire the stability of the electronic structure of noble gas. The shared electron pair is common to the two atoms and holds them together.
In a simple covalent bond, each atom brings an electron to the bond, that is, a pair of electrons is shared between two atoms.
In a double covalent bond, each atom brings two electrons to the bond, that is, two pairs of electrons are shared between two atoms.
Finally, in a triple covalent bond, each atom brings three electrons to the bond, that is, three pairs of electrons are shared between two atoms.
Then, a single covalent bond is formed when two atoms share a pair of electrons.
Ответ:
Ionic Bonding.
Explanation:
The bond here is in between two ions: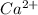 and
and 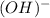
Since both are ions, this is an ionic bond.