Cold water (cp = 4.18 kj/kg⋅°c) leading to a shower enters a well-insulated, thin-walled, double-pipe, counterflow heat exchanger at 10°c at a rate of 0.95 kg/s and is heated to 70°c by hot water (cp = 4.19 kj/kg⋅°c) that enters at 85°c at a rate of 1.6 kg/s. determine (a) the rate of heat transfer and (b) the rate of entropy generation in the heat exchanger.
Solved
Show answers
More tips
- F Food and Cooking How to Make Sushi: A Step-by-Step Guide to Perfectly Rolled Delights...
- C Cities and Countries Which Country has the Most Expensive Visa?...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
Answers on questions: Physics
- E English What’s the relationship between the characters in macbeth you...
- M Mathematics Plz and explain as much as possible show work...
- H History The new monarchs after King James II included Mary of the House of...
- M Mathematics Naomi pays $1.50 each time she rides the city bus. She has already ridden the bus 18 times so far this year. The expression 1.50b+1.50(18) represents the amount...
- P Physics 3) A man holds a tennis ball (mass 58.5*10-3 kg) 2 meters off the ground and drops it. How long does it take the tennis ball to hit the ground...

Ответ:
(a) The rate of heat transfer is 238.26 kW.
(b) Rate of entropy generation is 0.063 kW/K.
Explanation:
(a) Taking the heat gained by the cold water into consideration,
Q = (0.95 kg/s)(4.18 kJ/kg.⁰C)(70-10) = 238.26 kW.
(b) Entropy generation = entropy in cold water + entropy in hot water
But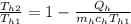
∴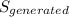 = (0.95)(4.18)(ln(343/283)) + (1.6)(4.19)(ln(1- (238.26/((1.6)(4.19)(358))))
= (0.95)(4.18)(ln(343/283)) + (1.6)(4.19)(ln(1- (238.26/((1.6)(4.19)(358))))
Ответ:
A
Explanation:
it would start as a low population which is smaller than get larger