dhhdjejdjdbdhdne
07.09.2019 •
Physics
Derive the velocity of an electron in a hydrogen atom using bohr's model. the electron has a mass of m, charge of (-e) and an orbit radius of r. make sure you explain all the teps including quantization of angular momentum to get full credits
Solved
Show answers
More tips
- W Work and Career Secrets of a Super Supervisor: Proper Team Management...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
Answers on questions: Physics
- E English 4. PART B: Which quote from paragraph 11 best supports the answer to Part A?...
- C Chemistry My everest is 29035 feet above sea level mr.kilimanjaro is 9700 feet lower how high is mr.kilimanjaro...
- M Mathematics Please I need a answer for this. As you guys can theirs two graphs which represent one sort of date , and the question is basically asking if it would good to show this date...
- E English What is the linking verb in this sentence? the sky looks clear and bright. a.bright b.sky c.looks d.clear...
- H History Pls help me helep help...

Ответ:
Explanation:
According to Bohr's Theory,
The force of attraction acting between the electron and the nucleus is equal to the centrifugal force acting on the electron.
Thus,
Where, Z is the atomic number or the number of protons
r is the atomic radius
v is the velocity of the electron
Also,
Accoriding to Bohr, the angular momentum is quantized. He states that the angular momemtum is equal to the integral multiple of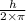 .
.
solving r from equation 2, we get that:
Putting in 1 , we get that:
Applying values for hydrogen atom,
Z = 1
Mass of the electron (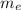 ) is 9.1093×10⁻³¹ kg
) is 9.1093×10⁻³¹ kg
Charge of electron (e) is 1.60217662 × 10⁻¹⁹ C
h is Plank's constant having value = 6.626×10⁻³⁴ m² kg / s
We get that:
Ответ: