During a family car trip, a family travels for 1 hour at 70 miles per hour. Then the family travels for 2 more hours at 55 miles per hour. What is the family's average speed during the trip?
A. 58.5 mph
B. 65 mph
C. 62.5 mph
D. 60 mph
Solved
Show answers
More tips
- G Goods and services Is an Air Ionizer Necessary for Your Home?...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Physics
- E English It wasn’t a stretch to say that Albert felt like a mouse in a maze. What form of figurative language is used in the above statement?...
- B Biology The model of human origins that attempts to incorporate all lines of evidence, including fossil and DNA evidence, is called the...
- M Mathematics Please help I need the answers 1-12...

Ответ:
Complete Question
The complete question is shown on the first uploaded image
The force on the lid when the air inside the cooker had been heated to 120°C is =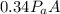
The force on the lid when the air inside the cooker had been heated to 20°C is =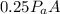
The
Explanation:
The main concept used to solve this problem is Pressure-Temperature Law.
Initially, find the pressure inside the pressure cooker by using the Pressure-Temperature Law. Then, find the force exerted by the air inside cooker on the lid of the cooker.
Fundamentals :
The Pressure-Temperature Law states that the pressure of the of a given gas held at constant volume is directly proportional to the Kelvin temperature. The increase in pressure results in an increase in the temperature and vice-versa.
The expression of the Pressure-Temperature Law is as follows:
Here,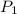 and
and 
are the initial and final pressures and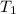 and
and 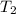
are the initial and final temperatures respectively.
The force in terms of pressure can be given by the following expression.
F=(ΔP) A
Here, A is the area on which the pressure is applied and ΔP is the change in pressure
The step- step calculation is shown on the second,third,fourth, fifth image
Note: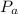 is the pressure of atmosphere
is the pressure of atmosphere