How many moles of a gas sample are in 2.5 l container at 373 k and 203 kpa?
Solved
Show answers
More tips
- G Goods and services How to Choose a Humidifier? Helpful Tips and Recommendations...
- W Work and Career What is the Most In-Demand Profession in the Modern World?...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Physics
- P Physics I need help pleaseee...
- P Physics Define , gravitational acceleration...
- P Physics Aforce is always needed to keep an object moving. a. true b. false...
- P Physics Why is the prime meridian located where it is ?...
- P Physics Experimentally, the free oscillation mode frequency is obtained by a read out from the oscilloscope screen. a student has found that 5 full periods of the damped oscillations cover...
- P Physics Consider the spring-mass-damper system discussed in class. the desired system response to a step input is that 2% ts ≤ 5 s, and os% ≤ 5%. a. sketch the region of the pole locations...
- P Physics (a) a 22.0-kg child is riding a playground merry-go-round that is rotating at 40.0 rev/min. what centripetal force is exerted if he is 1.25 m from its center? (b) what centripetal...
- P Physics Can turgor pressure be used to illustrate and demonstrate the effect of force and motion in a plant?...
- P Physics Acar drives to the right. there is a large amount of air resistance, and the cars engine provides the cars forward motion. which force on the diagram below represents the weight of...
- P Physics What are the units for speed velocity and acceleration?...

Ответ:
Explanation:
The given data is as follows.
P = 203 k Pa, V = 2.5 L
T = 373 K, n = ?
R = 0.0821 atm L/ K mol
Convert pressure from k Pa to atm as follows.
Now, using the ideal gas equation we will calculate the number of moles as follows.
PV = nRT
n =
=
=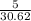 mol
mol
= 0.163 mol
Thus, we can conclude that number of moles in the given gas sample is 0.163 mol.
Ответ:
1. Type of Material
2. Charge of an object
3. movement of an object
4. Presence of an electric field
Hope this helps uwu <3