In a line graph comparing two variables, the independent variable is plotted along this axis
Solved
Show answers
More tips
- S Style and Beauty How to Choose the Perfect Hair Color?...
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Physics
- P Physics Which activity enable humans to produce new genetic combinations in other organisms...
- P Physics Where do coaster cars have the least amount of potential energy?...
- P Physics What is the minimum horizontal force needed to start a 300. kilogram steel block on a steel table in motion?...
- C Chemistry Determining Potehtlal Ehergy Complete these sentences. If a bouncing ball has a total energy of 20 J and a kinetic energy of 5 J, the ball s potential energy is If the kinetic energy...
- H History I need help! Anything helps...
- E English How does a simile most clearly contribute to the meaning of these lines from The Wreck of the Hesperus, by Henry Wadsworth Longfellow...
- E English What famous American writer doubted Shakespeare authorship? What was the point of his example of catfish fishing? PL HELP...
- M Mathematics HELP ASAP 2 MINUTES find the volume of a cylinder with a radius or 3.14...
- P Physics What is another name for Kepler s Third Law? Select all that apply.A) law of motionB) law of periodsC) law of harmoniesD) law of universal gravitation...
- M Mathematics Jing is helping the kindergarten teacher put together bags of crayons. Each bag has 4 crayons, and 1/4 of the crayons in each bag are green. What fraction of the crayons in 3 bags...

Ответ:
Explanation: Using ideal gas equation:
PV = nRT
P= pressure = 3.5 atm
V= volume =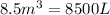
n = no of moles = 1900
R= gas constant =0.0821 Latm\molK
R= gas constant = 8.314 J/Kmol
T= temperature = 190.7 K
M= molecular mass of nitrogen = 28 g/mol