salvadorperez26
01.10.2019 •
Biology
Identify the process you would predict to have an increase in entropy?
a. condensation of water
b. freezing water
c. melting ice
d. deposition of co2 (changing from a gas to solid)
Solved
Show answers
More tips
- A Auto and Moto How to Start a Diesel Engine in Cold Weather?...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
- C Computers and Internet How Do You Refill Cartridges?...
Answers on questions: Biology
- C Chemistry A bottle with 18.02 grams of water contains 6.0221 x 10^23 molecules of water. A ream of paper is 2.00 inches thick and contains 500.0 pieces of paper. If a stack of paper...
- B Biology When the land and the sea warm up at different rates during the day, what is the most likely result? a. a breeze b. rain c.snow d. warm temperature...
- P Physics If an atom has 17 protons, 14 neutrons, and 21 electrons, what is the atom s electrical charge? uwu . ( type like this if you want to) : 0...
- B Biology The diagram shows how light energy from the sun drives the processes of photosynthesis and cellular respiration. which statement best describes the relationship between...
- B Biology Which of the following sources would be most likely to have reliable data...

Ответ:
Entropy is increases anytime it makes a bigger mess or creates disorderly. Entropy is decreases anytime it cleans up a mess. For example, condensing mist in the air into one droplet organizes the mess and that would be negative entropy. I hope this is making sense.
Ответ:
Ответ:
B
Explanation:
Isotopes are elements which have same atomic number but different mass number.
General representation of an element is given as: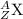
where,
Z represents Atomic number
A represents Mass number
X represents the symbol of an element
Thus for helium , which is shown to be
Mass number is defined as the sum of number of protons and neutrons that are present in an atom.
Mass number = Number of protons + Number of neutrons = 4
Number of neutrons = 4-2 =2
Atomic number is defined as the number of protons or number of electrons that are present in an atom.
Atomic number = Number of electrons = Number of protons = 2
Thus the representation with 2 protons and 2 neutrons will be the most common isotope of helium.