1.33*10^ 23 molecules of carbon dioxide to moles
Solved
Show answers
More tips
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry If an object has a density of 8.65 g/cm3, what is its density in units of kg/m3?...
- C Chemistry Does polonium lend or borrow electrons?...
- C Chemistry If an aqueous solution of is added to a mixture of pb2+ and ba2+, the lead ion will precipitate, but the barium ion will remain in solution. a. naoh b. na2so4 c....
- C Chemistry Combination reactions always forms what?...
- C Chemistry Manganese reacts with sulfuric acid to form manganese sulfate and hydrogen. what are the products in this reaction nitrogen manganese and sulfuric acid sulfuric...
- M Mathematics It s time to put it all together! Now that you know how to construct, it s your turn to create your own construction. Using your compass and straightedge, complete...
- M Mathematics How many more quarts is 10 quarts than 6 pints...
- C Chemistry How many moles of ammonia are produced when 6 moles of hydrogen gas react with nitrogen gas?...
- E English 100 POINTS!! Pls hurry it’s timed Read the excerpt from Joseph Chamberlain s speech I Believe in a British Empire. I cannot look forward without dread to handing...
- P Physics How to produce radio wave?...

Ответ:
0.22moles of carbon dioxide
Explanation:
Given parameters:
Number of molecules of carbon dioxide = 1.33 x 10²³molecules
Unknown:
Number of moles = ?
Solution:
To solve this problem, we use the relationship below:
1 mole of a substance contains 6.02 x 10²³moleccules;
So;
1.33 x 10²³molecules will contain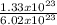 = 0.22moles of carbon dioxide
= 0.22moles of carbon dioxide
Ответ:
Total mass of the compound:=18.20+2.45
=20.65
Percentage by mass of hydrogen:=2.45÷20.65×100
=11.86%
Explanation:
Just follow the steps i have provided above