betru12315
20.03.2020 •
Chemistry
1. Using the Nernst equation, calculate the equilibrium potential for Ca21 and for Cl2 from the following sets of data: a. Given [Ca21]o 5 1 mM, [Ca21]i 5 100 nM, find ECa21 b. Given [Cl2]o 5 110 mM, [Cl2]i 5 10 mM, find ECl2
Solved
Show answers
More tips
- H Health and Medicine How to Treat Whooping Cough in Children?...
- L Leisure and Entertainment Unlocking the Secrets of Fast and Effective Tectonic Learning...
- A Animals and plants Уход за джунгариками: полезные советы и рекомендации...
- S Style and Beauty How to knit a hooded cowl?...
- S Style and Beauty How to Break in New Shoes: 7 Simple Methods...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- A Art and Culture Attention, the Final Episode of Margo is Almost Here!...
- H Health and Medicine Novomin: What is it and how to use it?...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...

Ответ:
Answer : The value of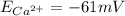 and
and 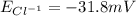
Explanation :
To calculate the equilibrium potential we are using Nernst equation:
where,
z = valency of ion
Part a :
Given:
z = valency of ion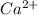 = 2
= 2
Part b :
Given:
z = valency of ion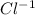 = 1
= 1
Ответ:
you could send the zoom I'd with password