xXCoryxKenshinXx
04.10.2021 •
Chemistry
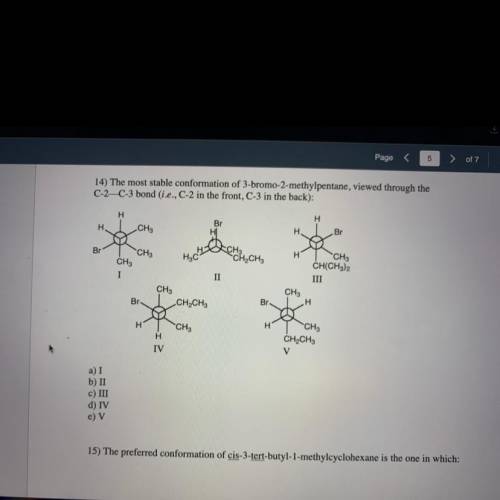
14) The most stable conformation of 3-bromo-2-methylpentane, viewed through the
C-2—C-3 bond (i.e., C-2 in the front, C-3 in the back):
H
H
Br
H
CH3
H.
Br
Br
CH
CH2CH3
CH3
CH3
H3C
CH3
I
CH3
Br
CH2CH3
II
H
CH(CH3)2
III
CH3
H
Br
A
H
CH3
H
H
IV
CH3
CH2CH3
V
a) I
b) II
c) III
d) IV
e) V
15 TL
Solved
Show answers
More tips
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry In addition to running aspartic acid, phenylalanine, aspartame and hydrolyzed aspartame, let s assume that you also ran leucine on your TLC plate in this lab. Would you expect...
- C Chemistry Which of the following would most likely result in an increase in reaction rate? placing the reactants on a hotplate placing the reactants in an ice bath placing the products...
- C Chemistry An aqueous magnesium chloride solution is made by dissolving 6.92 6.92 moles of MgCl 2 MgCl2 in sufficient water so that the final volume of the solution is 2.00 L 2.00 L ....
- C Chemistry Technician A says that water floats to the top of fuel in the tank. Technician B says that water is more likely to be drawn into the system when the fuel level is low. Which...
- C Chemistry Find the H+ concentration of of a solution that has a pH solution of 12.24...
- C Chemistry Consider the following initial rate data (at 273 K) for the decomposition of a substrate (substrate 1) which decomposes to product 1 and product 2: [Substrate 1] (M) Initial...
- C Chemistry You have studied the gas-phase oxidation of HBr by O2: 4 HBr(g) + O2(g) → 2 H2O(g) + 2 Br2(g) You find the reaction to be first order with respect to HBr and first order with...
- C Chemistry Draw the arrow-pushing mechanism of a generic esterification reaction: b) From the spectral data (NMR, IR, MS) you were given, identify the structure of your ester product....
- C Chemistry We have a lost in comet then you thick of...
- C Chemistry 3500 J of energy are added to a 0.5 mol sample of iron at 293 K. What is the final temperature of the iron in kelvins? The molar heat of iron is 25.1 J/(mol•K)....

Ответ:
Kp = 929551.4
Explanation:
First of all, we state the equilibrium:
4PBr₃ (g) ⇄ P₄(g) + 6Br₂(g)
In order to determine Kp, we need the partial pressure of each gas at equilibrium. Expression for Kp is:
{(Parial Pressure P₄) . (Partial Pressure Br₂)⁶} / (Partial Pressure PBr₃)⁴
Kp = 99.2 . 97.2⁶ / 97.4⁴
Kp = 929551.4
Take account that Kp can be also calculated from Kc.
Kp = Kc (RT)^Δn where Δn is the value for (final moles - initial moles) of any gas