ineedhelp2285
01.08.2019 •
Chemistry
15. a 22.4-l sample of which of the following substances, at stp, would contain 6.02 x 10^23 representative particles? *a. goldb. sodium chloridec. sulfurd. carbon dioxide16. what information is needed to calculate the percent composition of a compound? *a. the formula of the compound and the atomic mass of its elementsb. the formula of the compound and its volumec. the weight of the sample to be analyzed and its molar volumed. the weight of the sample to be analyzed and its state of matter
Solved
Show answers
More tips
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
Answers on questions: Chemistry
- C Chemistry All group 1 metals have similar reactions with water. explain why, in terms of electrronic structure PLEASE ANSWER AND PLEASE EXPLAINWILL AWARD THE FIRST AND CORRECR ANSWER WITH 30...
- C Chemistry Which of these is an example of a connection between two different Earth systems? ice melting on a lake deer drinking from a stream a robin hatching from an egg minerals forming under...
- C Chemistry Pls help me I will give brainliest first answer i have 20 more minutes left of this quiz so pretty pls will you help me! By how much did carbon dioxide levels increase from 1980 to...
- C Chemistry Help pls true or false 1. Oxygen from the lungs moves into the blood by active transport. 2. Osmosis requires no ATP energy 3. Active transport over time results in equilibrium 4....
- C Chemistry What information does the potential energy diagram reveal about a reaction?...
- C Chemistry Which statement summarizes the law of superposition? A. The oldest rock is found in the bottom layer of a rock formation. B. The rarest elements are found in the lowest layer of a...
- C Chemistry Which formula is comparing a sample of a gas under two different conditions of volume and temperature, if the pressure is constant? ⚪︎ V1 V2 ____ = ___ T1 T2 ⚪︎ xy = k ⚪︎ V __ = k...
- C Chemistry How are the atomic number and the number of protons related to each other?...
- M Mathematics With algebra what are the possible numbers of positive real, negative real, and complex zeros of f(x) = 4x3 + x2 + 10x − 14? positive real: 1 negative real: 0 complex: 2 positive...
- B Biology The water table rises and falls with precipitation is the bottom layer of the saturated zone is a layer of porous rock below an aquifer always stays in the same place...

Ответ:
The answer for 15 is oxygen or carbon dioxide they are the same thing!
The answer for 16 is the formula of the compound and the atomic mas of its elements!
This is for if you take connections!
Ответ:
Answer :
Part 15 : Only gold contains number of particles.
number of particles.
Part 16 : The correct option is, (a) the formula of the compound and the atomic mass of its elements.
Explanation :
Part 15 :
As we know that, 1 mole of substance occupies 22.4 L volume of gas and 1 mole of substance contains number of particles.
number of particles.
So, 22.4 L volume of gas contains number of particles.
number of particles.
(a) Gold :
Gold contains number of particles.
number of particles.
(b) Sodium chloride (NaCl) :
In sodium chloride, there are 2 atoms. So, it contains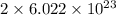 number of particles.
number of particles.
(c) Sulfur (S₈) :
In sulfur, there are 8 atoms. So, it contains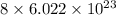 number of particles.
number of particles.
(d) Carbon dioxide (CO₂) :
In carbon dioxide, there are 3 atoms. So, it contains number of particles.
number of particles.
Hence, from this we conclude that only gold contains number of particles.
number of particles.
Part 16 :
Percent composition : It is calculated by dividing the mass of each element in one mole of the compound that means dividing the mass of each element by the total molar mass of the compound.
Formula used :
Hence, the information needed to calculate the percent composition of a compound is, the formula of the compound and the atomic mass of its elements.
Ответ:
good question